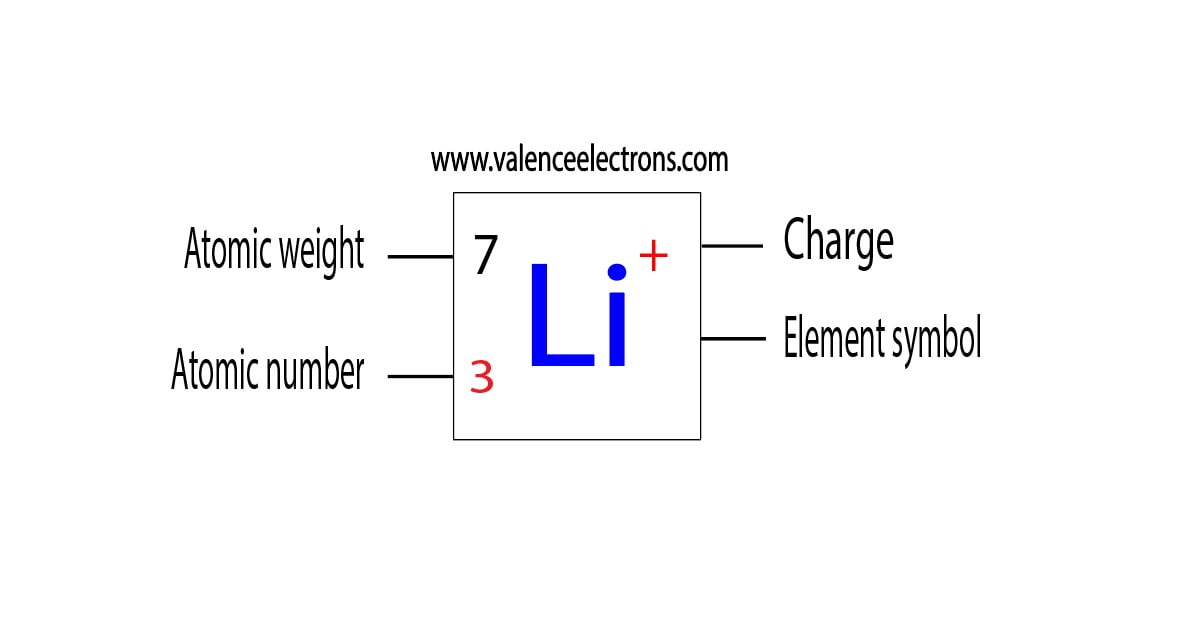
Lithium Electron Configuration and Orbital Diagram Model
Lithium is the 3rd element in the periodic table and the symbol is ‘Li’. Lithium has an atomic number of 3, which means that its atom has only 3 electrons outside its nucleus. To write the electron configuration for lithium, the first two electrons enter the 1s orbital. Since the 1s can hold only two electrons, the remaining electrons will enter the 2s orbital. Therefore, the electron configuration of lithium will be 1s2 2s1.
The electron configuration of lithium refers to the arrangement of electrons in the Lithium atom’s orbitals. It describes how electrons are distributed among the various atomic orbitals and energy levels, and provides a detailed map of where each electron is likely to be found.
To understand the mechanism of lithium electron configuration, you need to understand two things. These are orbits and orbitals. Also, you can arrange electrons in those two ways. In this article, I have discussed all the points necessary to understand the mechanism of lithium electron configuration. I hope this will be helpful in your study.
Electron arrangement for Lithium through Bohr Model
Scientist Niels Bohr was the first to give an idea of the atom’s orbit. He provided a model of the atom in 1913 and provided a complete idea of orbit in that model.
The electrons of the atom revolve around the nucleus in a certain circular path. These circular paths are called orbits (shells or energy levels). These orbits are expressed by n. [n = 1,2,3,4 . . . The serial number of the orbit]
The name of the first orbit is K, L is the second, M is the third, and N is the name of the fourth orbit. The electron holding capacity of each orbit is 2n2.
| Shell Number (n) | Shell Name | Electrons Holding Capacity (2n2) |
| 1 | K | 2 |
| 2 | L | 8 |
| 3 | M | 18 |
| 4 | N | 32 |
Explanation:
- Let, n = 1 for K orbit. So, the maximum electron holding capacity in the K orbit is 2n2 = 2 × 12 = 2 electrons.
- n = 2, for L orbit. The maximum electron holding capacity in the L orbit is 2n2 = 2 × 22 = 8 electrons.
- n=3 for M orbit. The maximum electron holding capacity in the M orbit is 2n2 = 2 × 32 = 18 electrons.
- n=4 for N orbit. The maximum electron holding capacity in N orbit is 2n2 = 2 × 42 = 32 electrons.
Therefore, the maximum electron holding capacity in the first shell is two, the second shell is eight and the 3rd shell can have a maximum of eighteen electrons.
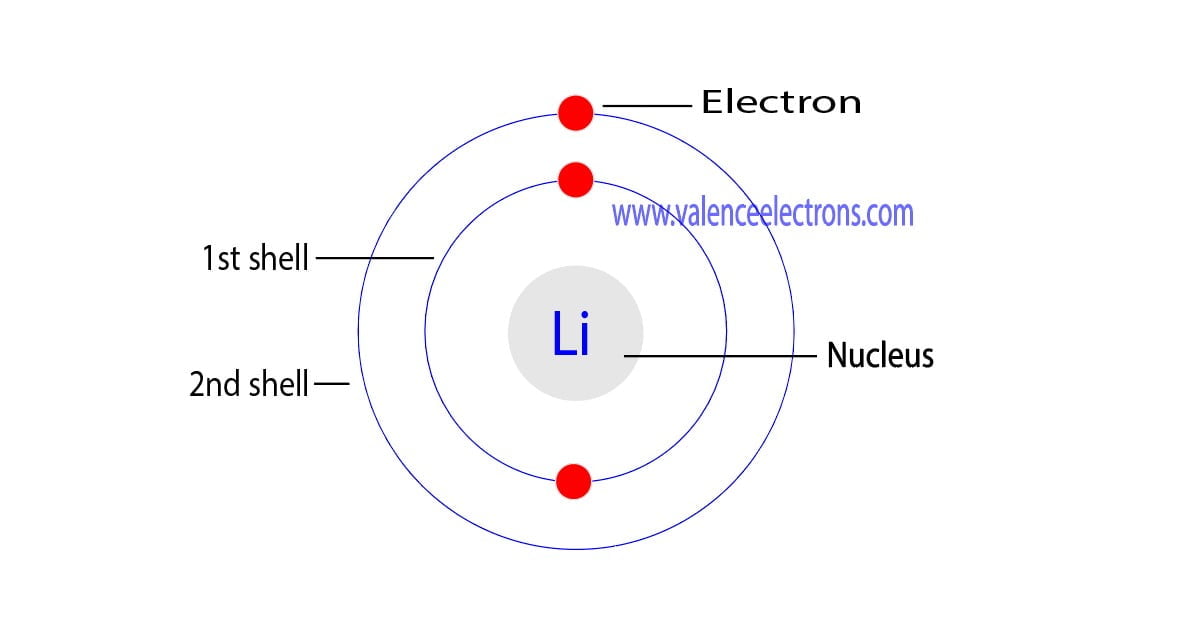
The atomic number is the number of electrons in that element. The atomic number of lithium is 3. That is, the number of electrons in lithium is 3. Therefore, a lithium atom will have two electrons in the first shell and one electron in the 2nd shell. Therefore, the order of the number of electrons in each shell of the lithium(Li) atom is 2, 1.
The Bohr atomic model has many limitations. In the Bohr atomic model, the electrons can only be arranged in different shells but the exact position, orbital shape, and spin of the electron cannot be determined.
Also, electrons can be arranged correctly from 1 to 18 elements. The electron arrangement of any element with atomic number greater than 18 cannot be accurately determined by the Bohr atomic model following the 2n2 formula. We can overcome all limitations of the Bohr model following the electron configuration through orbital.
Electron configuration of lithium through orbital
Atomic energy shells are subdivided into sub-energy levels. These sub-energy levels are also called orbital. The most probable region of electron rotation around the nucleus is called the orbital.
The sub-energy levels depend on the azimuthal quantum number. It is expressed by ‘l’. The value of ‘l’ is from 0 to (n – 1). The sub-energy levels are known as s, p, d, and f.
| Orbit Number | Value of ‘l’ | Number of subshells | Number of orbitals | Subshell name | Electrons holding capacity | Electron configuration |
| 1 | 0 | 1 | 1 | 1s | 2 | 1s2 |
| 2 | 0 1 | 2 | 1 3 | 2s 2p | 2 6 | 2s2 2p6 |
| 3 | 0 1 2 | 3 | 1 3 5 | 3s 3p 3d | 2 6 10 | 3s2 3p6 3d10 |
| 4 | 0 1 2 3 | 4 | 1 3 5 7 | 4s 4p 4d 4f | 2 6 10 14 | 4s2 4p6 4d10 4f14 |
Explanation:
- If n = 1,
(n – 1) = (1–1) = 0
Therefore, the value of ‘l’ is 0. So, the sub-energy level is 1s. - If n = 2,
(n – 1) = (2–1) = 1.
Therefore, the value of ‘l’ is 0, 1. So, the sub-energy levels are 2s, and 2p. - If n = 3,
(n – 1) = (3–1) = 2.
Therefore, the value of ‘l’ is 0, 1, 2. So, the sub-energy levels are 3s, 3p, and 3d. - If n = 4,
(n – 1) = (4–1) = 3
Therefore, the value of ‘l’ is 0, 1, 2, 3. So, the sub-energy levels are 4s, 4p, 4d, and 4f. - If n = 5,
(n – 1) = (n – 5) = 4.
Therefore, l = 0,1,2,3,4. The number of sub-shells will be 5 but 4s, 4p, 4d, and 4f in these four subshells it is possible to arrange the electrons of all the elements of the periodic table.
| Sub-shell name | Name source | Value of ‘l’ | Value of ‘m’ (0 to ± l) | Number of orbital (2l+1) | Electrons holding capacity 2(2l+1) |
| s | Sharp | 0 | 0 | 1 | 2 |
| p | Principal | 1 | −1, 0, +1 | 3 | 6 |
| d | Diffuse | 2 | −2, −1, 0, +1, +2 | 5 | 10 |
| f | Fundamental | 3 | −3, −2, −1, 0, +1, +2, +3 | 7 | 14 |
The orbital number of the s-subshell is one, three in the p-subshell, five in the d-subshell, and seven in the f-subshell. Each orbital can have a maximum of two electrons.
The sub-energy level ‘s’ can hold a maximum of two electrons, ‘p’ can hold a maximum of six electrons, ‘d’ can hold a maximum of ten electrons, and ‘f’ can hold a maximum of fourteen electrons.
Aufbau is a German word, which means building up. The main proponents of this principle are scientists Niels Bohr and Pauli. The Aufbau method is to do electron configuration through the sub-energy level.
The Aufbau principle is that the electrons present in the atom will first complete the lowest energy orbital and then gradually continue to complete the higher energy orbital.
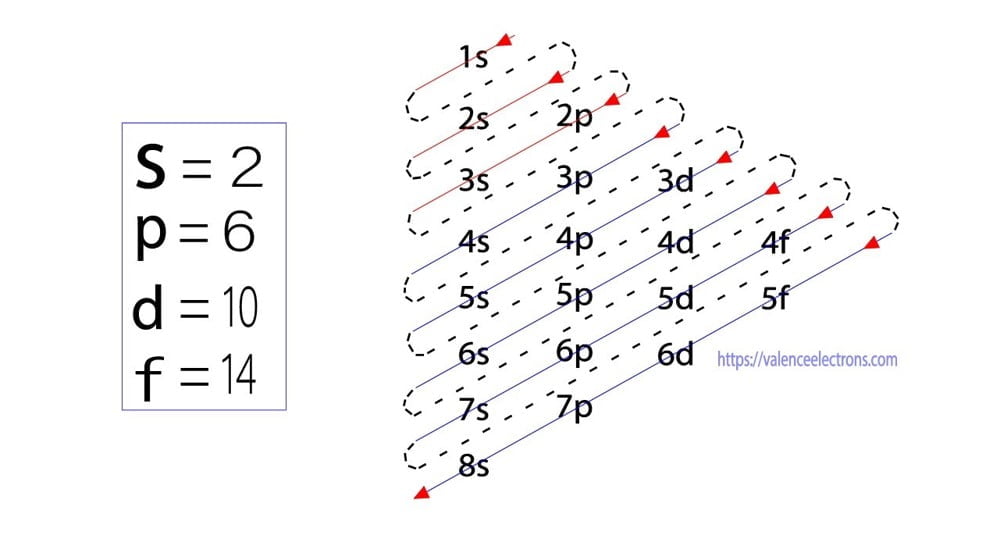
The energy of an orbital is calculated from the value of the principal quantum number ‘n’ and the azimuthal quantum number ‘l’. The orbital for which the value of (n + l) is lower is the low energy orbital and the electron will enter that orbital first.
| Orbital | Orbit (n) | Azimuthal quantum number (l) | Orbital energy (n + l) |
| 3d | 3 | 2 | 5 |
| 4s | 4 | 0 | 4 |
Here, the energy of 4s orbital is less than that of 3d. So, the electron will enter the 4s orbital first and enter the 3d orbital when the 4s orbital is full. Following the Aufbau principle, the sequence of entry of electrons into orbitals is 1s 2s 2p 3s 3p 4s 3d 4p 5s 4d 5p 6s 4f 5d 6p 7s 5f 6d 7p.
Therefore, the complete electron configuration for lithium should be written as 1s2 2s1.
Note: The unabbreviated electron configuration of lithium is [He] 2s1.
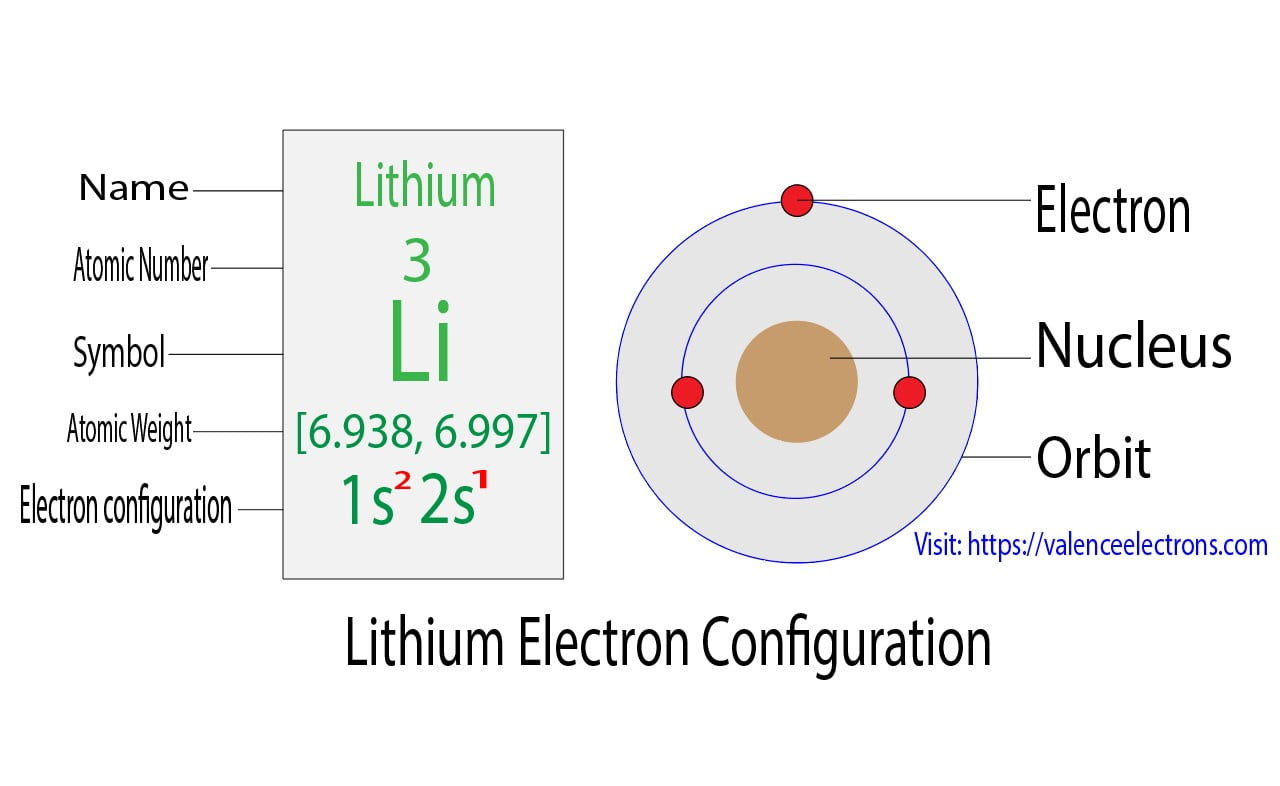
How to draw the orbital diagram of lithium
The orbital diagram of lithium is a graphical representation of the electron configuration of lithium atom. This diagram shows how the electrons in the lithium atom are arranged in different orbitals and indicates the spin of electrons. Orbital is the region of space around the nucleus of an atom where electrons are found.
To draw an orbital diagram of any atom, you first need to know the atomic orbitals and the orbital notation for that atom, which is already discussed above. Also, you need to know Hund’s and Paul’s exclusion principle. These two fundamental principles explain how these electrons occupy the 1s and 2s orbitals. Below, we define each principle and apply it to lithium, helping students understand its atomic structure and reactivity.
Hund’s Rule: Hund’s Rule states that when electrons fill orbitals of the same energy (called degenerate orbitals, like the three 2p orbitals), they spread out to occupy each orbital singly before pairing up. Additionally, these single electrons have the same spin (e.g., all spin-up, ↑) to minimize electron-electron repulsion and maximize stability.
Pauli Exclusion Principle: The Pauli Exclusion Principle states that no two electrons in an atom can have the same set of four quantum numbers. In practice, this means each orbital can hold a maximum of two electrons, and these electrons must have opposite spins (e.g., one ↑ and one ↓).
Orbital diagrams are usually represented by boxes. Each box represents an orbital and the arrows within the box represent the position of the electron. The boxes are arranged in order of energy of the orbitals. The lowest energy orbitals are closest to the nucleus and the higher energy orbitals are progressively further away from the nucleus in order of their energy levels.
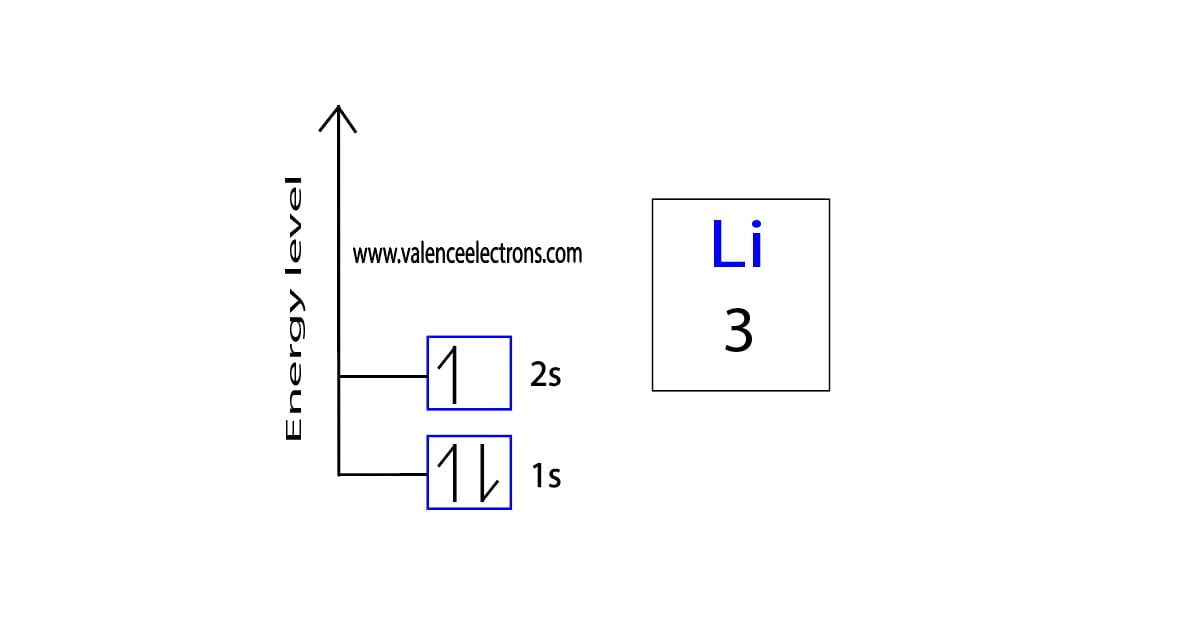
To write the orbital diagram of lithium, the 1s orbital is the closest and lowest energy orbital to the nucleus. Therefore, the electrons will first enter the 1s orbital. According to Hund’s principle, the first electron will enter the 1s orbital in the clockwise direction and the second one will enter the 1s orbital in the anti-clockwise direction. The 1s orbital is now filled with two electrons. So the remaining one electron will enter the 2s orbital in the clockwise direction. This is clearly shown in the figure of the orbital diagram of lithium.
Why This Matters for Lithium
Electron configuration for Lithium-ion (Li+)
After the electron configuration, the last shell of the lithium atom has an electron. In this case, the valence electrons of lithium are one, and also valency is 1.
The elements that have 1, 2, or 3 electrons in the last shell donate the electrons in the last shell during bond formation. The lithium atom donates an electron of the last shell to turn into a lithium-ion (Li+).
The element that forms a bond by donating electrons is called cation. Lithium donates an electron and turns into a positive ion. Therefore, lithium is a cation element.
Li – e– → Li+
Here, the electron configuration of lithium ion(Li+) is 1s2. This electron configuration shows that the lithium ion(Li+) acquired the electron configuration of helium and it achieves a stable electron configuration.
Determine the group and period of lithium through the electron configuration
The last orbit of an element is the period of that element. The electron configuration shows that the last shell of the lithium is 2. So, the period of the lithium atom is 2.
On the other hand, the number of electrons present in the last orbit of an element is the number of groups in that element. An electron exists in the last orbit of the lithium atom.
That is, the group number of lithium is 1. Therefore, we can say that the period of the lithium is 2 and the group is 1.
Determine the block of lithium through the electron configuration
If the last electron enters the s-orbital after the electron configuration of the element, then that element is called the s-block element. The electron configuration shows that the last electron of lithium enters the s-orbital. Therefore, lithium is the s-block element.
The elements in group 2 are the s-block elements. And helium is the s-block element. There are 14 s-block elements in the 118 elements of the periodic table.
Why lithium is an alkali metal?
The elements in group 1 of the periodic table are alkali metals. Lithium is the element of group number 1 of the periodic table. Therefore, lithium is an alkali metal. (Excluding the only hydrogen)
Oxidation and Reduction Properties
The element of group 1 is lithium. Which is an intense reducing element. The lithium atom donates an electron of the s-orbital. The lithium atom forms the electron structure of the helium element. The intensity of lithium is so high that lithium atoms reduce the hydrogen to form lithium hydride compounds.
2Li + H2 → 2LiH
The ionization potential of the alkali metal decreases as it moves from top to bottom within the group of periodic tables. That is, the reduction capacity continues to increase.
As such, lithium is a weak reducing agent and cesium is a severe reducing agent. However, lithium is the most powerful reducing agent among alkali metals. The oxidizing potential of lithium atoms is +3.04.
Properties of Lithium
- The atomic number of lithium is 3. The atomic number of an element is the number of electrons in that element. Therefore, the number of electrons in lithium is three.
- Lithium’s standard atomic weight is 6.941.
- Lithium is an alkali and an intensely negative metal. Its oxides and hydroxides are strong alkalis.
- The value of electronegativity of lithium atoms is comparatively much lower. The value of electronegativity of lithium atoms is 0.98.
- The covalent radius of the lithium atom is 128 ± 7 pm.
- The ionic radius of the lithium atom is 6.0 × 10 –2 nm.
- Lithium atom van der Waals radius 182 pm.
- Ionization energies of lithium atoms E1 = 520 kj/mol, E 2 = 7298 kj/mol, E3 = 11815 kj/mol.
- The oxidation states of lithium atoms are +1.
- The volume of lithium atoms is 13.1cc / mol.
- The melting point of a lithium atom is 180.50 ° C (453.65 K, 356.90 ° F). And the boiling point is 1342 C.
- The period of the lithium element is 2. And the group is 1.
- The number of valency and valence electrons of a lithium atom is 1.
- Lithium forms both covalent and ionic bonds.
- Lithium is a highly electrically positive element. As a result, the lithium atom is stable and produces ions in the solution.
- An electron exists in the last orbit of lithium.
- The atomic radius of a lithium atom is 152 pm.
- Lithium atoms react with hydrogen, oxygen, and halogen to form compounds.
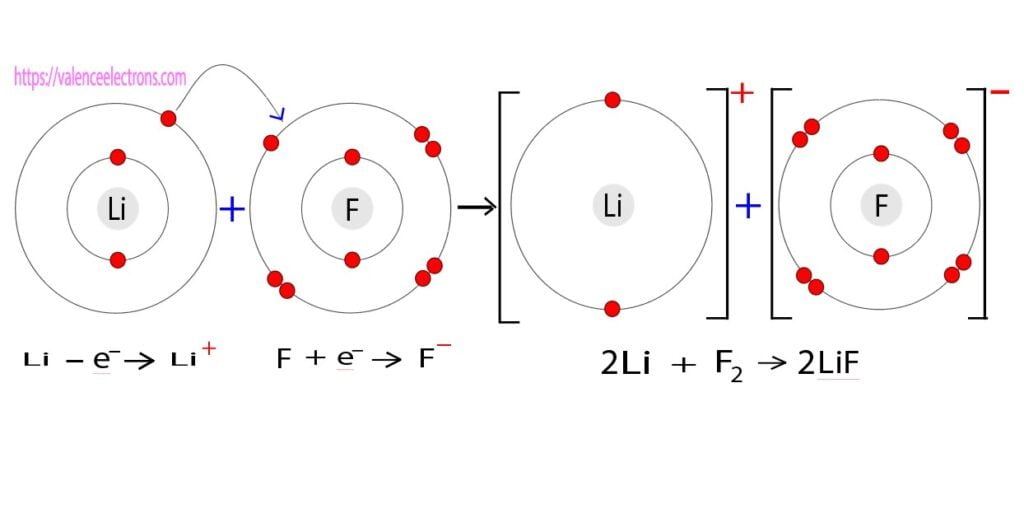
Ionic bond of lithium
Lithium atoms form an ionic bond with fluorine atoms through the exchange of electrons. An electron exists in the last orbit of the lithium atom. The lithium atom wants to be more stable by forming one helium atom by eliminating an electron in the last orbit.
On the other hand, The electron configuration of fluorine atom shows that the seven electrons exist in the last orbit. The fluorine atom wants to be more stable like the neon atom by accepting an electron. The lithium atom donates an electron of its last orbit to the fluorine atom and LiF forms compounds through ionic bonds.
lithium compound formation
Reaction of lithium with halogen
Lithium atoms react with halogen to form halide compounds.
- 2Li + F2 → 2LiF
- 2Li + Cl2 → 2LiCl
- 2Li + Br2 → 2LiBr
- 2Li + I2 → 2LiI
The activity of alkali metals with specific halogens gradually increases from lithium to cesium. That is, Li < Na < K < Rb < Cs. But except for lithium, all other alkali metal halides dissolve very easily in water.
The intense gravitational force between Li+ and F– ion in the crystal reduces the solubility of LiF. Therefore, LiF is less soluble in water. LiCl, LiBr, and LiI are soluble in organic solvents ethanol, and propanol.
Reaction of oxygen
Group-1 elements, such as alkali metals, have a greater ability to bind to oxygen. Lithium (an alkali metal) reacts with controlled amounts of oxygen to form LiO (oxide) compounds.
4Li (s) + O2 (g) → 2Li2O (lithium oxide)
Heating lithium with excess oxygen produces the compound Li2O2.
2Li + O2 → Li2O2 (lithium peroxide).
Reaction of hydrogen
Alkali metals react with dry hydrogen at a temperature of about 400 ° C to form metallic hydrides. However, the lithium atom is different from all other alkali metals. Lithium reacts with hydrogen at a temperature of 800°C to produce lithium hydride.
2Li + H2 → 2 LiH.
The reaction activity decreases as it moves from lithium to cesium element. Metal hydride stability tends to decrease from LiH to CsH.
Lithium oxide
Lithium reacts with water to produce LiOH.
2Li + 2H2O → 2LiOH + H2
Lithium is oxidized by oxygen in the air to produce alkaline Li2O.
4Li + O2 → 2Li2O
Li2O reacts with water to produce mild alkali LiOH.
Li2O + H2O → 2LiOH
As Li2O is alkaline, it destroys the acidity of HCl and produces salt and water.
Li2O + 2HCl → 2LiCl + H2O
Reaction of lithium with other elements
Lithium atoms react with sulfur and phosphorus to form sulfide and phosphide compounds.
16 Li + S8 → 8Li2S
12Li + P4 → 4Li3P
The alkali metal lithium reacts with nitrogen(N2) at high temperatures to form nitride compounds.
6Li + N2 → 2Li3N
Exceptional properties of the lithium atom
In general, elements of the same group have similarities in physical and chemical properties. The elements of group 1 are alkali metals. The elements are lithium(Li), sodium(Na), potassium(K), rubidium(Rb), cesium(Cs).
However, the properties of lithium are different from the properties of all the other elements in this group. The elements in group 1 form ionic bonds in halide compounds. However, the halide compounds of the lithium atom have covalent bonds.
For this reason, sodium halide is soluble in water but lithium halide is very slightly soluble in water. LiCl, LiBr, and LiI are soluble in various organic solvents (ethanol, propanol, ether).
Alkali metals react with hydrogen at 400° C to form hydride compounds. But lithium atoms form lithium hydride compounds with hydrogen at a temperature of 800° C.
2Li + H2 (800° C) → 2LiH.
The reasons for the exceptional properties of lithium (Li) are-
- The small size of Li atom and Li+ ion.
- The polarization capacity of Li+ ions is high.
- The zero d orbital is missing at the valence level.
- Lithium is a high ionic potential element.
Due to the exceptional properties of lithium, several distinctions of lithium with other alkali metals have been observed. They are-
- Lithium is less active than alkali metals. Its bright color does not fade easily when it comes in contact with air. In the case of other alkali metals, their bright color fades in contact with air.
- Lithium (Li) reacts with oxygen to form only monoxide (Li2O) compounds. But never forms peroxide (Li2O2) or superoxide (LiO2).
- Lithium is the only alkali metal that reacts with nitrogen (N2) to produce Li3N.
6Li + N2 → 2Li3N.
All other alkali metals do not react with nitrogen (N2). - When the alkaline metal nitride is heated, it decomposes, and metallic nitride and oxygen are produced.
2MNO3 → 2 MNO2 + O2. Here, [M = Na, K, Rb] - When lithium nitride is heated, it decomposes and produces lithium monoxide (Li2O), NO2, and oxygen (O2).
4LiNO3 (heat) → 2Li2O + 4NO2 + O2 - Hydroxides of alkali metals have a strong base. However, lithium hydroxide is a weak base.
- Lithium chloride (LiCl) combines with water to form dihydrate compounds. But NaCl, KCl, and CsCl never form hydrate compounds.
Reference
- Gole, J. L., Childs, R. H., Dixon, D. A., & Eades, R. A. (1980). The electronic structure of the lithium trimer anion and cation. The Journal of Chemical Physics, 72(12), 6368-6375.
- Ivanov, M. V., & Schmelcher, P. (1998). Ground state of the lithium atom in strong magnetic fields. Physical Review A, 57(5), 3793.
- Stacey, W., & Marsiglio, F. (2012). Why is the ground-state electron configuration for lithium 1s22s?. Europhysics Letters, 100(4), 43002.
Great explanation of lithium’s electron configuration! I love how you broke down the orbital diagram. It really helps clarify the concept for those of us who find these topics challenging. Looking forward to your next post!
Great explanation of lithium’s electron configuration! I particularly liked how you illustrated the orbital diagram—it makes the concept much clearer. It’s fascinating to see how the electron arrangement influences its chemical properties. Thanks for the insights!
Great post! I found the explanation of lithium’s electron configuration and the orbital diagram especially helpful in visualizing how electrons are arranged. It’s fascinating how the simple structure of lithium can lead to such diverse chemical behavior. Thanks for breaking it down so clearly!
This post on lithium’s electron configuration and orbital diagram model is super helpful! I appreciate how clearly you explained the placement of electrons and the significance of orbital shapes. It really deepened my understanding of atomic structure. Thank you for such an informative read!
Great explanation of lithium’s electron configuration and orbital diagram! It’s fascinating to see how the arrangement of electrons influences its chemical properties. I appreciate the clear visuals in the post that made it easier to understand the concept. Thank you for this informative article!
Great explanation of lithium’s electron configuration! I found the orbital diagram really helpful in visualizing how the electrons are arranged. It’s fascinating to see how this setup influences lithium’s chemical properties. Thank you for breaking it down so clearly!