Sodium Electron Configuration and Atomic Orbital Diagram
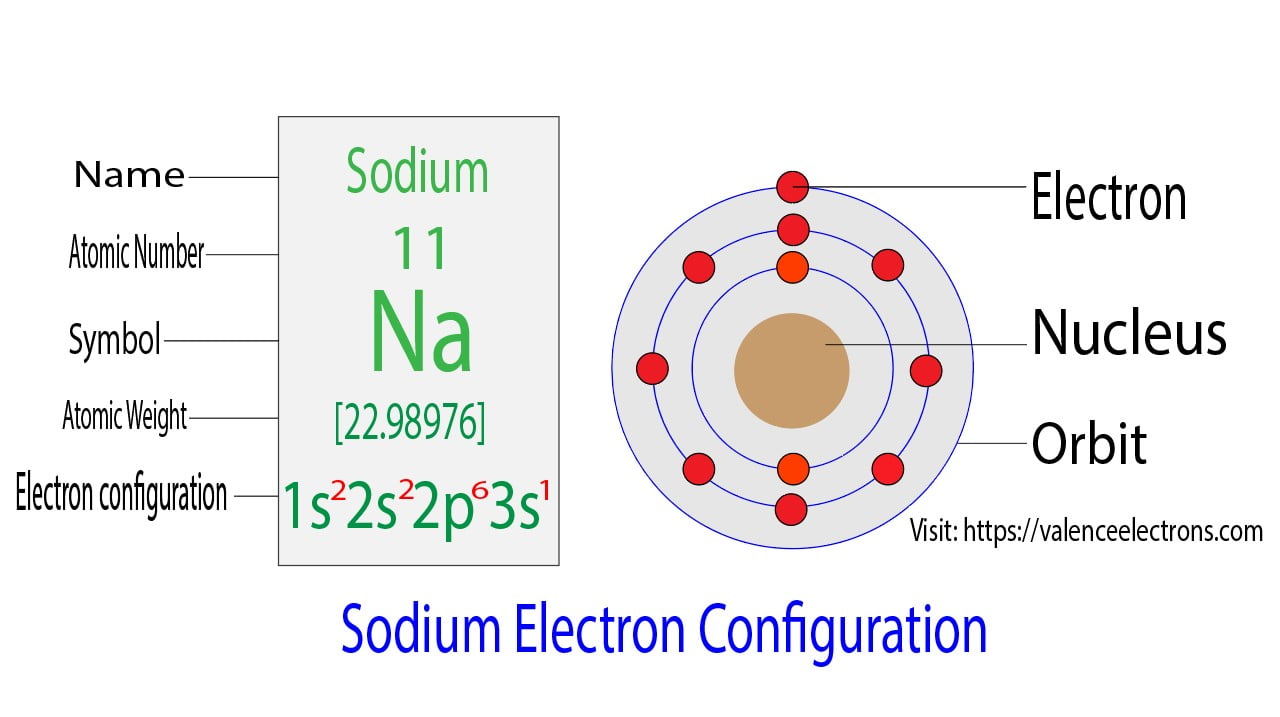
Sodium is the 11th element in the periodic table and the symbol is ‘Na’. Sodium’s atomic number is 11, which means that its atom has eleven electrons around its nucleus.
To write the electron configuration for sodium, the first two electrons enter the 1s orbital. Since the 1s orbital can hold only two electrons the next two enter the 2s orbital. The next six electrons enter the 2p subshell. The p subshell can hold a maximum of six electrons. Hence, the remaining one electron enters the 3s orbital. Therefore, the electron configuration of sodium will be 1s2 2s2 2p6 3s1.
The electron configuration of sodium refers to the arrangement of electrons in the sodium atom’s orbitals. It describes how electrons are distributed among the various atomic orbitals and energy levels, and provides a detailed map of where each electron is likely to be found.
To understand the mechanism of sodium electron configuration, you need to understand two basic things. These are orbits and orbitals. Also, you can arrange electrons in those two ways. In this article, I have discussed all the necessary points to understand the mechanism of sodium electron configuration. I hope this will be helpful in your study.
Electron arrangement for Sodium through orbit
Scientist Niels Bohr was the first to give an idea of the atom’s orbit. He provided a model of the atom in 1913 and provided a complete idea of orbit in that model.
The electrons of the atom revolve around the nucleus in a certain circular path. These circular paths are called orbits (shells or energy levels). These orbits are expressed by n. [n = 1,2,3,4 . . . The serial number of the orbit]
The name of the first orbit is K, L is the second, M is the third, and N is the name of the fourth orbit. The electron holding capacity of each orbit is 2n2.
| Shell Number (n) | Shell Name | Electrons Holding Capacity (2n2) |
| 1 | K | 2 |
| 2 | L | 8 |
| 3 | M | 18 |
| 4 | N | 32 |
Explanation:
- Let, n = 1 for K orbit. So, the maximum electron holding capacity in the K orbit is 2n2 = 2 × 12 = 2 electrons.
- n = 2, for L orbit. The maximum electron holding capacity in the L orbit is 2n2 = 2 × 22 = 8 electrons.
- n=3 for M orbit. The maximum electron holding capacity in the M orbit is 2n2 = 2 × 32 = 18 electrons.
- n=4 for N orbit. The maximum electron holding capacity in N orbit is 2n2 = 2 × 42 = 32 electrons.
Therefore, the maximum electron holding capacity in the first shell is two, the second shell is eight and the 3rd shell can have a maximum of eighteen electrons.
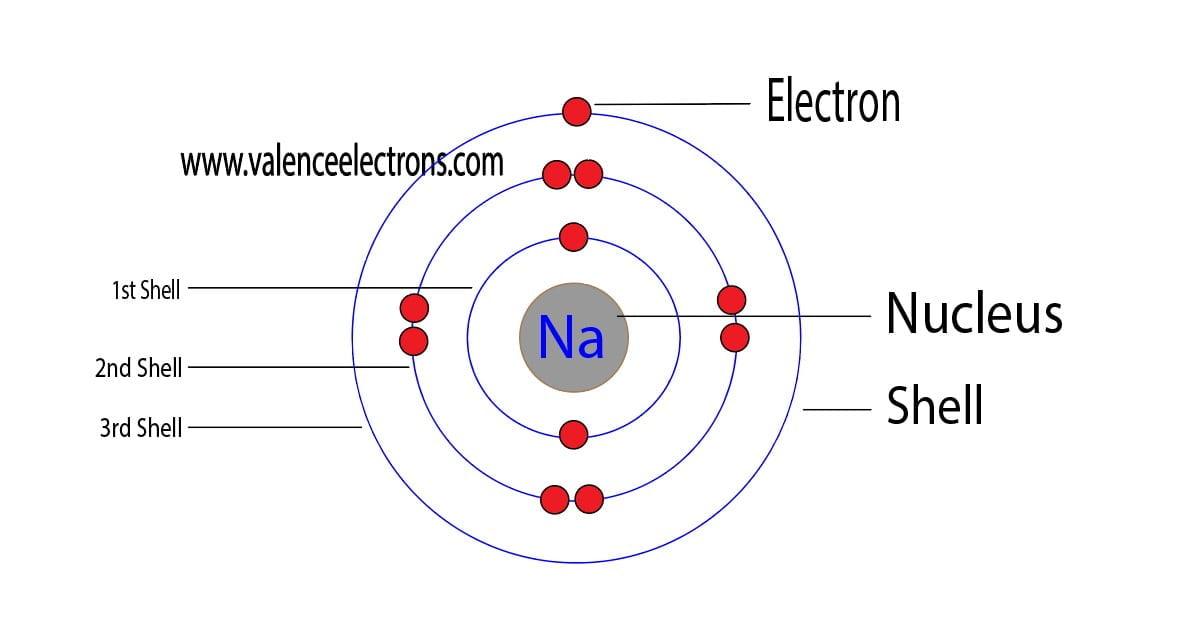
The atomic number is the number of electrons in that element. The atomic number of sodium is 11. That is, the number of electrons in sodium is 11. Therefore, a sodium atom will have two electrons in the first shell, eight in the 2nd orbit, and an electron in the 3rd shell. Hence, the order of the number of electrons in each shell of the sodium atom is 2, 8, 1.
The Bohr atomic model has many limitations. In the Bohr atomic model, the electrons can only be arranged in different shells but the exact position, orbital shape, and spin of the electron cannot be determined.
Also, electrons can be arranged correctly from 1 to 18 elements. The electron arrangement of any element with atomic number greater than 18 cannot be accurately determined by the Bohr atomic model following the 2n2 formula. We can overcome all limitations of the Bohr model following the electron configuration through orbital.
Electron configuration of Sodium through orbital
Atomic energy shells are subdivided into sub-energy levels. These sub-energy levels are also called orbital. The most probable region of electron rotation around the nucleus is called the orbital.
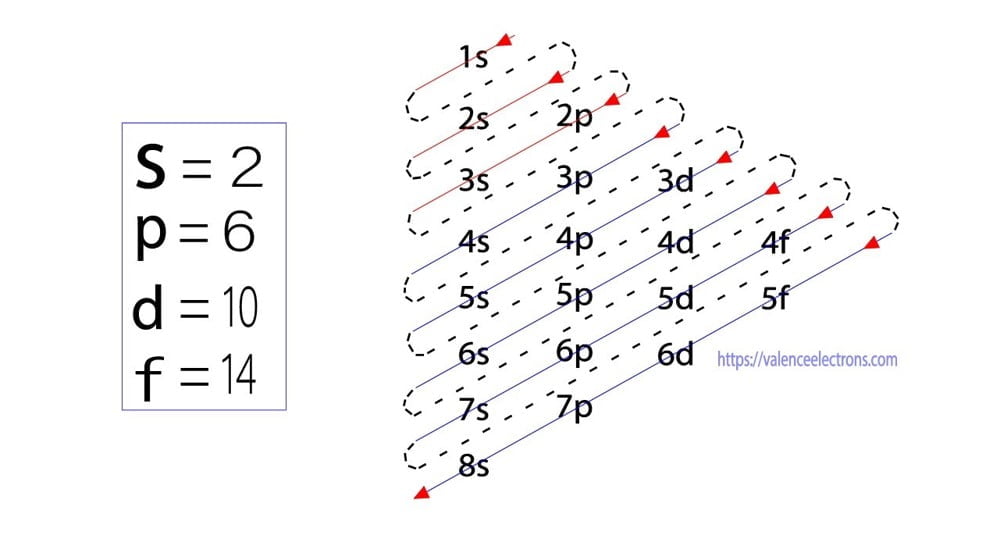
The sub-energy levels depend on the azimuthal quantum number. It is expressed by ‘l’. The value of ‘l’ is from 0 to (n – 1). The sub-energy levels are known as s, p, d, and f.
| Orbit Number | Value of ‘l’ | Number of subshells | Number of orbitals | Subshell name | Electrons holding capacity | Electron configuration |
| 1 | 0 | 1 | 1 | 1s | 2 | 1s2 |
| 2 | 0 1 | 2 | 1 3 | 2s 2p | 2 6 | 2s2 2p6 |
| 3 | 0 1 2 | 3 | 1 3 5 | 3s 3p 3d | 2 6 10 | 3s2 3p6 3d10 |
| 4 | 0 1 2 3 | 4 | 1 3 5 7 | 4s 4p 4d 4f | 2 6 10 14 | 4s2 4p6 4d10 4f14 |
Explanation:
- If n = 1,
(n – 1) = (1–1) = 0
Therefore, the value of ‘l’ is 0. So, the sub-energy level is 1s. - If n = 2,
(n – 1) = (2–1) = 1.
Therefore, the value of ‘l’ is 0, 1. So, the sub-energy levels are 2s, and 2p. - If n = 3,
(n – 1) = (3–1) = 2.
Therefore, the value of ‘l’ is 0, 1, 2. So, the sub-energy levels are 3s, 3p, and 3d. - If n = 4,
(n – 1) = (4–1) = 3
Therefore, the value of ‘l’ is 0, 1, 2, 3. So, the sub-energy levels are 4s, 4p, 4d, and 4f. - If n = 5,
(n – 1) = (n – 5) = 4.
Therefore, l = 0,1,2,3,4. The number of sub-shells will be 5 but 4s, 4p, 4d, and 4f in these four subshells it is possible to arrange the electrons of all the elements of the periodic table.
| Sub-shell name | Name source | Value of ‘l’ | Value of ‘m’ (0 to ± l) | Number of orbital (2l+1) | Electrons holding capacity 2(2l+1) |
| s | Sharp | 0 | 0 | 1 | 2 |
| p | Principal | 1 | −1, 0, +1 | 3 | 6 |
| d | Diffuse | 2 | −2, −1, 0, +1, +2 | 5 | 10 |
| f | Fundamental | 3 | −3, −2, −1, 0, +1, +2, +3 | 7 | 14 |
The orbital number of the s-subshell is one, three in the p-subshell, five in the d-subshell, and seven in the f-subshell. Each orbital can have a maximum of two electrons.
The sub-energy level ‘s’ can hold a maximum of two electrons, ‘p’ can hold a maximum of six electrons, ‘d’ can hold a maximum of ten electrons, and ‘f’ can hold a maximum of fourteen electrons.
Aufbau is a German word, which means building up. The main proponents of this principle are scientists Niels Bohr and Pauli. The Aufbau method is to do electron configuration through the sub-energy level.
The Aufbau principle is that the electrons present in the atom will first complete the lowest energy orbital and then gradually continue to complete the higher energy orbital.
The energy of an orbital is calculated from the value of the principal quantum number ‘n’ and the azimuthal quantum number ‘l’. The orbital for which the value of (n + l) is lower is the low energy orbital and the electron will enter that orbital first.
| Orbital | Orbit (n) | Azimuthal quantum number (l) | Orbital energy (n + l) |
| 3d | 3 | 2 | 5 |
| 4s | 4 | 0 | 4 |
Here, the energy of 4s orbital is less than that of 3d. So, the electron will enter the 4s orbital first and enter the 3d orbital when the 4s orbital is full. Following the Aufbau principle, the sequence of entry of electrons into orbitals is 1s 2s 2p 3s 3p 4s 3d 4p 5s 4d 5p 6s 4f 5d 6p 7s 5f 6d 7p.
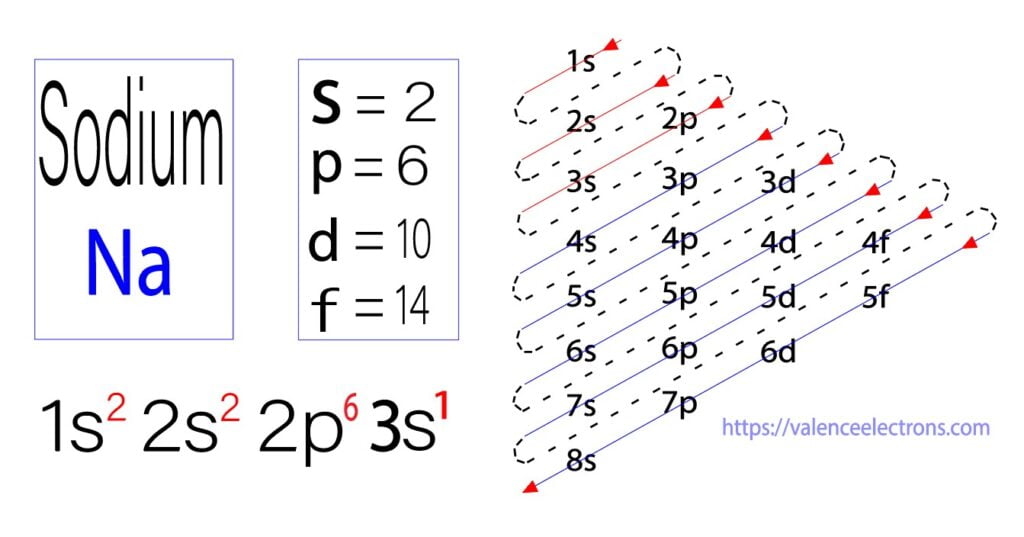
Therefore, the complete electron configuration for sodium should be written as 1s2 2s2 2p6 3s1.
Note: The unabbreviated electron configuration of sodium is [Ne] 3s1. When writing an electron configuration, you have to write serially.
How to draw the orbital diagram of sodium?
Orbital diagrams are usually represented by boxes. Each box represents an orbital and the arrows within the box represent the position of the electron. The boxes are arranged in order of energy of the orbitals. The lowest energy orbitals are closest to the nucleus and the higher energy orbitals are progressively further away from the nucleus in order of their energy levels. To write the orbital diagram of sodium, you have to write the orbital notation of sodium, which has been discussed in detail above.
The orbital diagram of sodium is a graphical representation of the electron configuration of a sodium atom. This diagram shows how the electrons in the sodium atom are arranged in different orbitals and indicates the spin of electrons. Orbital is the region of space around the nucleus of an atom where electrons are found.
The 1s orbital is the closest and lowest energy orbital to the nucleus. Therefore, the electrons will first enter the 1s orbital. According to Hund’s principle, the first electron will enter the 1s orbital in the clockwise direction and the second electron will enter the 1s orbital in the anti-clockwise direction.
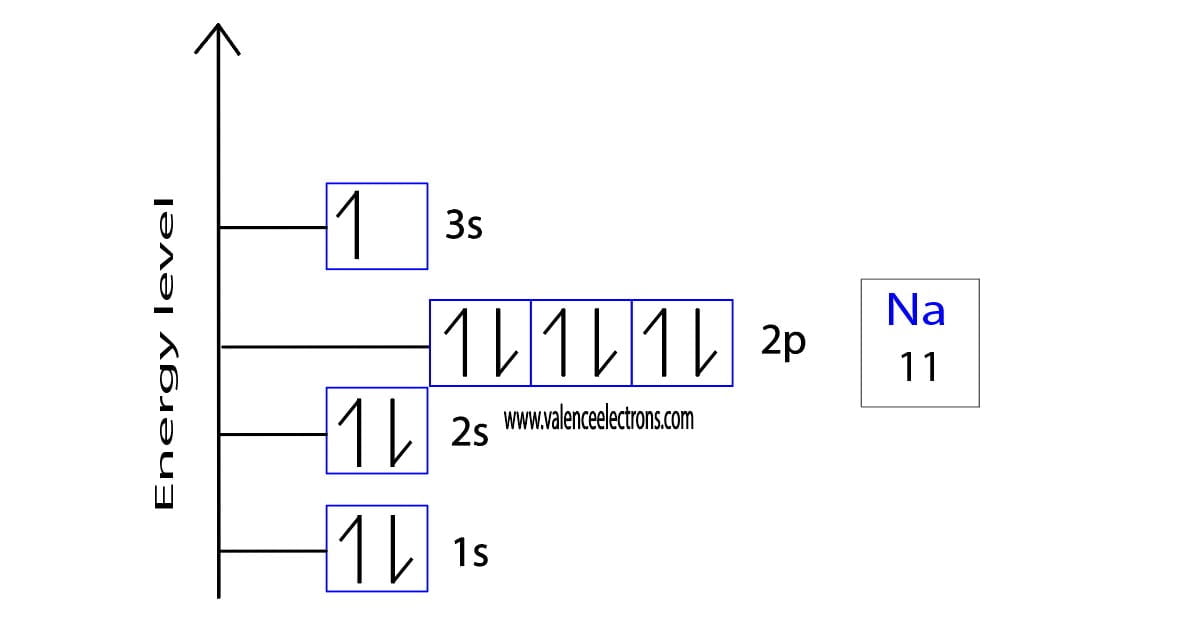
The 1s orbital is now filled with two electrons. The next two electrons will enter the 2s orbital, just like the 1s orbital. The next three electrons will enter the 2p (px, py, pz) orbital one by one in a clockwise direction, and the next three electrons will enter the 2p (px, py, pz) orbital one by one in a counterclockwise direction. The 2p orbital is now full of electrons. Then the remaining one electron will enter the 3s orbital in the clockwise direction. This is clearly shown in the figure of the orbital diagram of sodium.
Electron configuration for Sodium ion(Na+)
The ground-state electron configuration of sodium is 1s2 2s2 2p6 3s1. The elements that have 1, 2, or 3 electrons in the last shell donate the electrons in the last shell during bond formation.
The elements that form bonds by donating electrons are called cations. Sodium donates an electron of the last shell to form bonds and turns into a sodium ion(Na+). That is, sodium is a cation element.
Na – e– → Na+
The electron configuration of sodium ion(Na+) is 1s2 2s2 2p6. This electron configuration shows that sodium ion(Na+) has acquired the electron configuration of neon(Ne) and it achieves an octave full stable electron configuration.
How to determine the group and period of sodium through the electron configuration?
The last orbit of an element is the period of that element. The electron configuration of the sodium atom shows that the last orbit of the sodium atom is 3(3s1). So, the period of sodium is 3.
On the other hand, the number of electrons present in the last orbit of an element is the number of groups in that element. An electron exists in the last orbital(3s1) of sodium. That is, the group number of sodium is 1. Therefore, we can say that the period of the sodium element is 3 and the group is 1.
How to determine the block of sodium through the electron configuration?
The elements in the periodic table are divided into four blocks based on the electron configuration of the element. The block of elements is determined based on the electron configuration of the element.
If the last electron enters the s-orbital after the electron configuration of the element, then that element is called the s-block element. The electron configuration of sodium shows that the last electron of sodium enters the s-orbital. Therefore, sodium is an s-block element.
How to determine the valency and valence electrons of sodium through the electron configuration?
The ability of one atom of an element to join another atom during the formation of a molecule is called valency(valence). The number of unpaired electrons in the last orbit of an element is the valency of that element. The electron configuration shows that the last shell of sodium has an unpaired electron. Therefore, the valency of sodium is 1.
Again, the valency of the element can be determined in another way. If the last orbit of an element has 1,2,3 or 4 electrons, then the number of electrons in the last orbit is the valency of that element. From the sodium electron configuration, we can say that an electron exists in the last orbit of sodium. Therefore, the valency of sodium is 1.
Again, the number of electrons in the last orbit of an element, the number of those electrons is the valence electrons of that element. we see that an electron exists in the last orbit of sodium. Therefore, the valence electrons of sodium are one.
Ionic properties of sodium atom
There are two types of ionic properties. One is a cation and the other is an anion. The sodium atom exhibits cation properties. When a charge-neutral atom leaves an electron and turns into a positive ion, it is called a cation.
Although the electron strength of the last orbit of an atom is higher, the force of attraction on that electron by the positive charge of the nucleus is less effective.
As the force of attraction is less effective, electrons are removed from the last orbit. And the atom turns into a cation. The sodium atom leaves an electron in its last orbit and turns into a cation.
Na – e– → Na+
Bonding of sodium atom
Sodium atoms always form ionic bonds. Sodium atoms form ionic bonds by exchanging electrons with chlorine atoms. The electron configuration of a sodium atom indicates that there is an electron in the 3rd orbit of the sodium atom.
The sodium atom wants to be as stable as the inert element neon by leaving an electron in its last orbit.
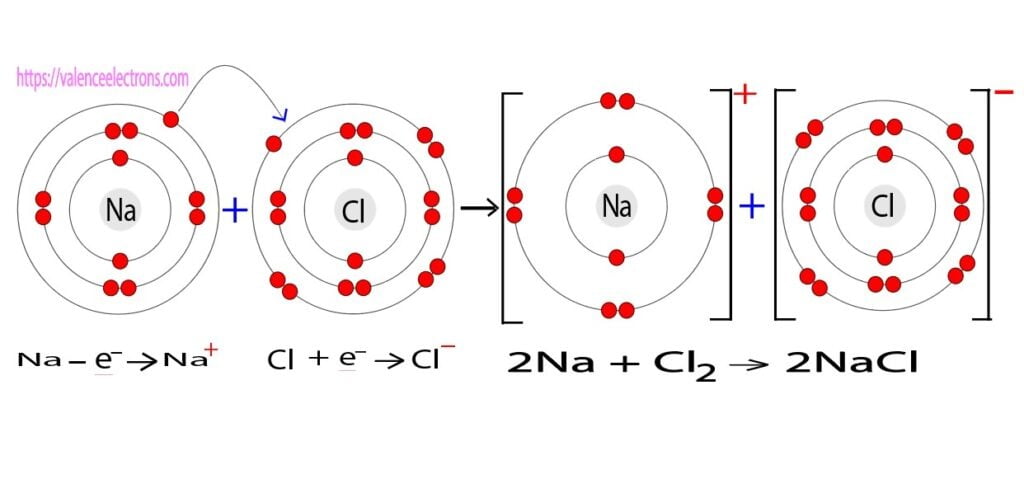
On the other hand, the electron configuration of a chlorine atom implies that the last orbit of a chlorine atom has seven electrons. The chlorine atom wants to fill the octave by taking an electron in its last orbit.
So, the sodium atom donates an electron of its last orbit to the chlorine atom. Sodium and chlorine atoms form sodium chloride(NaCl) compounds through ionic bonds by exchanging electrons in their last orbit.
2Na + Cl2 → 2NaCl (sodium chloride)
Reaction of sodium atom
Sodium is an alkali metal. Sodium forms bonds and compounds with various elements. Here are a few discussed.
Reaction of sodium atoms with halogen
Halogen elements are fluorine(F), chlorine(Cl), bromine(Br), iodine(I), and astatine(At). The s-block element is sodium. Sodium reacts with halogen to form halide compounds.
2Na + F2 → 2NaF (sodium fluoride)
2Na + Cl2 → 2NaCl (sodium chloride)
2Na + Br2 → 2NaBr (sodium bromide)
2Na + I2 → 2NaI (sodium iodide)
Reaction of sodium atom with oxygen
Sodium is an element of group-1. Therefore, the tendency of sodium atoms to bind to oxygen is much higher. Sodium atoms combine with oxygen to form oxides, dioxides, and super-oxides.
4Na + O2 (heat) → 2Na2O
Alkaline metal sodium is heated with excess oxygen to form sodium peroxide.
2Na + O2 → Na2O2 (sodium peroxide)
Reaction of sodium atom with water
The alkali metal is sodium. The reduction value of the sodium atom is very high. For this, the best reduction of hydrogen is the sodium atom. Sodium metal reacts with water to produce hydrogen gas.
2Na + 2H2O → 2NaOH + H2
Reaction of sodium atom with hydrogen
Sodium atoms react with dry hydrogen at 400 °C to form hydride compounds.
2Na + H2 (400 C) → 2NaH
Reaction of sodium atom with other elements
Sodium atoms react with sulfur and phosphorus to form compounds.
16Na + S8 (heat) → 8 Na2S
12Na + P4 (heat) → 4 Na3P
Again, sodium atoms react with mercury to produce amalgam.
Na + Hg → NaHg
Properties of sodium atom
- The atomic number of sodium atoms is 11. The atomic number of an element is the number of electrons and protons in that element. That is, the number of electrons and protons in the sodium atom is eleven.
- The sodium electron configuration is 1s2 2s2 2p6 3s1.
- The active atomic mass of the sodium atom is 22.98976928.
- Sodium is an alkali metal.
- The number of valency and valence electrons of a sodium atom is 1.
- Sodium atoms are the 3rd period of the periodic table and an element of the first group.
- Sodium is a highly electrically positive element. Therefore, sodium forms stable compounds.
- Sodium is a cation element.
- Sodium atoms form ionic bonds.
- The sodium atom can easily release 1 electron in its last orbit. For this reason, it is a very active metal.
- Sodium cation is filled by welded electrons. Therefore, its cation is diamagnetic and colorless.
- The melting point of a sodium atom is 370.944 K (97.794 °C, 208.029 °F). And the boiling point is 1156.090 K (882.940 °C, 1621.292 °F).
- The electronegativity of sodium atoms is 0.93 (Pauling scale).
- Sodium is an s-block element.
- The oxidation states of sodium are +1.
- The reduction potential of sodium atoms is 2.71.
- The atomic volume of the sodium atom is 27.7cc.
- Ionization energies of sodium atoms are 1st: 495.8 kJ/mol, 2nd: 4562 kJ/mol, 3rd: 6910.3 kJ/mol.
- The electron addiction of sodium atoms is –52.8kj/mol.
- The covalent radius of the sodium atom is 166±9 pm.
- The sodium atom van der Waals radius is 227 pm.