How Many Valence Electrons Does Sodium (Na) Have?
Sodium is the eleventh element in the periodic table. Sodium is an alkali metal and its symbol is ‘Na’. The valence electrons are the total number of electrons in the last orbit(shell).
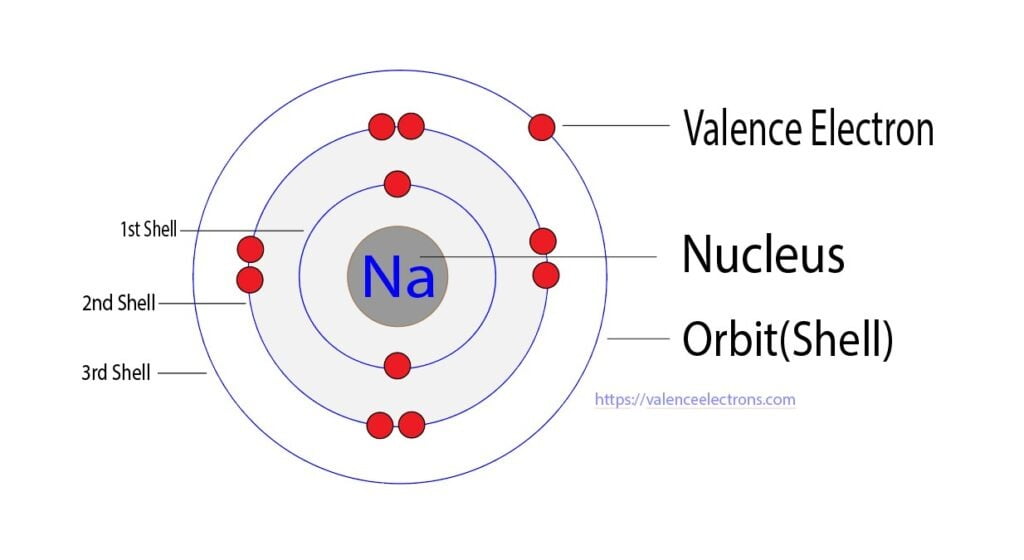
The total number of electrons in the last shell after the electron configuration of sodium is called the valence electrons of sodium. The last shell of sodium has an electron. Therefore, the valence electron of sodium is one. The valence electrons determine the element’s properties and participate in forming bonds. Sodium atoms participate in the formation of bonds through their valence electrons.
How do you calculate the number of valence electrons in a sodium atom?
The valence electron has to be determined by following a few steps. The electron configuration is one of them. It is not possible to determine the valence electron without electron configuration. Knowing the electron configuration in the right way, it is very easy to determine the valence electrons of all the elements.
However, valence electrons can be easily identified by arranging electrons according to the Bohr principle. Now we will learn how to determine the valence electrons of sodium(Na).
Step-1: Determining the total number of electrons in sodium
First, we need to know the total number of electrons in the sodium(Na) atom. To know the number of electrons, you need to know the number of protons in sodium. To know the number of protons, you need to know the atomic number of the sodium element.
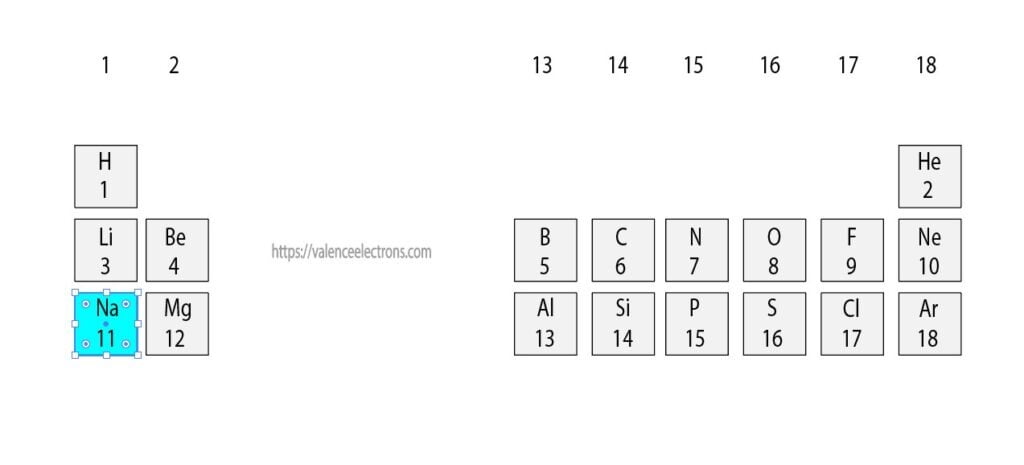
To know the atomic number we need to take the help of a periodic table. It is necessary to know the atomic number of sodium elements from the periodic table. The atomic number is the number of protons and electrons equal to protons located outside the nucleus.
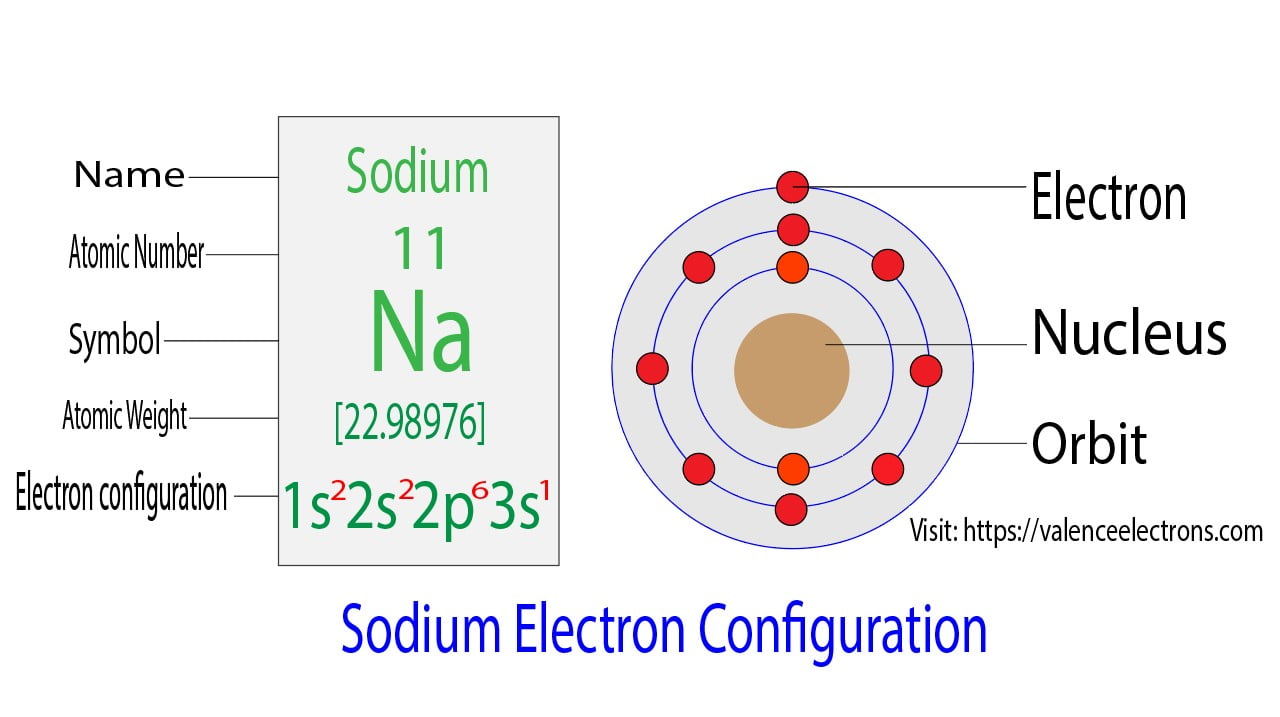
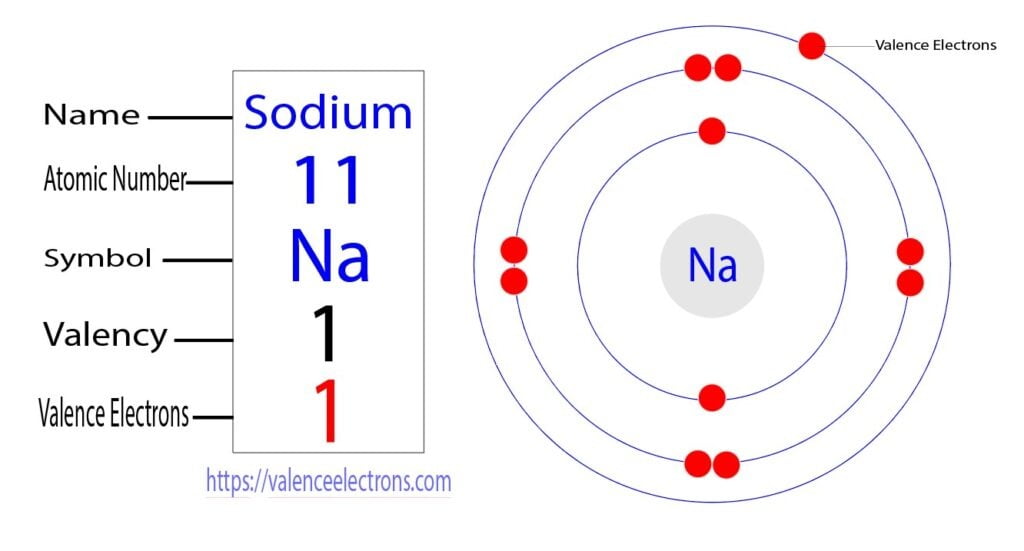
That is, we can finally say that there are electrons equal to the atomic number in the sodium atom. From the periodic table, we see that the atomic number of sodium is 11. That is, a sodium atom has a total of eleven electrons.
Step-2: Need to do electron configuration of sodium
Step 2 is very important. In this step, the electrons of sodium have to be arranged. We know that sodium atoms have a total of eleven electrons. The electron configuration of sodium shows that there are two electrons in the K shell, eight in the L shell, and one in the M shell(orbit). That is, the first shell of sodium has two electrons, the second shell has eight electrons and the 3rd shell(orbit) has one electron.
Step-3: Determine the valence shell and calculate the total electrons
The third step is to diagnose the valence shell(orbit). The last shell after the electron configuration is called the valence shell. The total number of electrons in a valence shell is called a valence electron. The electron configuration of sodium shows that the last shell of sodium has an electron. Therefore, the valence electrons of sodium are one.
What is the valency of sodium?
The ability of one atom of an element to join another atom during the formation of a molecule is called valency(valence). The number of unpaired electrons in the last orbit of an element is the valency of that element. The electron configuration of sodium shows that there is an unpaired electron in the last orbit of sodium. Therefore, the valency of sodium is 1.
How many valence electrons does sodium ion(Na+) have?
The elements that have 1, 2, or 3 electrons in the last shell donate the electrons in the last shell during bond formation. The elements that form bonds by donating electrons are called cations. That is, sodium is a cation element. Sodium donates the electron of the last shell to form bonds and turns into a sodium ion(Na+).
Na – e– → Na+
The electron configuration of sodium ion(Na+) is 1s2 2s2 2p6. The electron configuration shows that sodium ion(Na+) have only two shells and the last shell has eight electrons. The electron configuration shows that the sodium atom has acquired the electron configuration of neon. That is, in this case, the valency of the sodium ion is +1. Since the last shell of a sodium ion has eight electrons, the valence electrons of sodium ion(Na+) are eight.
Compound formation of Sodium
Sodium participates in the formation of bonds through its valence electron. We know that the valence electron in sodium is one. This valence electron participates in the formation of bonds with atoms of other elements.
The electron configuration of chlorine shows that the valence electrons of chlorine are seven. The sodium atom donates its valence electron to the chlorine atom and the chlorine atom receives that electron.
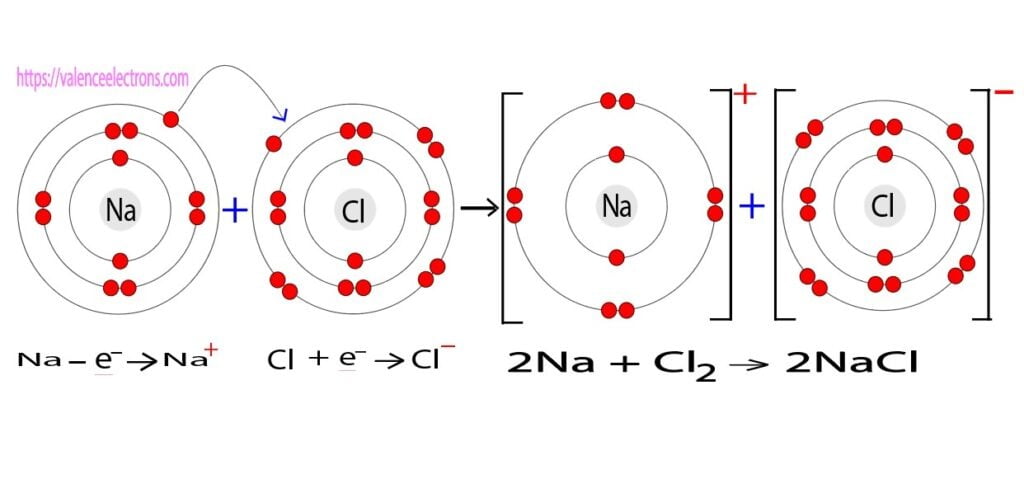
As a result, chlorine acquires the electron configuration of argon and, the sodium atom acquires the electron configuration of neon. Chlorine and sodium atoms form sodium chloride(NaCl) bonds through electron exchange. Sodium chloride(NaCl) is ionic bonding.