Helium Electron Configuration and Orbital Diagram Model
Helium is the 2nd element in the periodic table and the symbol is ‘He’. The atomic number of helium is 2, which means its atom has only 2 electrons around the nucleus. To write the electron configuration for helium, the first two electrons enter the 1s orbital. Hence, the electron configuration for helium will be 1s2.
The electron configuration of helium refers to the arrangement of electrons in the helium atom’s orbitals. It describes how electrons are distributed among the various atomic orbitals and energy levels, and provides a detailed map of where each electron is likely to be found.
To understand the mechanism of helium electron configuration, you need to understand two things. These are orbits and orbitals. Also, you can arrange electrons in those two ways. In this article, I have discussed all the points necessary to understand the mechanism of helium electron configuration. I hope this will be helpful in your study.
Electron arrangement for Helium through shell
Scientist Niels Bohr was the first to give an idea of the atom’s orbit. He provided a model of the atom in 1913 and provided a complete idea of orbit in that model.
The electrons of the atom revolve around the nucleus in a certain circular path. These circular paths are called orbits (shells or energy levels). These orbits are expressed by n. [n = 1,2,3,4 . . . The serial number of the orbit]
The name of the first orbit is K, L is the second, M is the third, and N is the name of the fourth orbit. The electron holding capacity of each orbit is 2n2.
| Shell Number (n) | Shell Name | Electrons Holding Capacity (2n2) |
| 1 | K | 2 |
| 2 | L | 8 |
| 3 | M | 18 |
| 4 | N | 32 |
Explanation:
- Let, n = 1 for K orbit. So, the maximum electron holding capacity in the K orbit is 2n2 = 2 × 12 = 2 electrons.
- n = 2, for L orbit. The maximum electron holding capacity in the L orbit is 2n2 = 2 × 22 = 8 electrons.
- n=3 for M orbit. The maximum electron holding capacity in the M orbit is 2n2 = 2 × 32 = 18 electrons.
- n=4 for N orbit. The maximum electron holding capacity in N orbit is 2n2 = 2 × 42 = 32 electrons.
Therefore, the maximum electron holding capacity in the first shell is two, the second shell is eight and the 3rd shell can have a maximum of eighteen electrons.
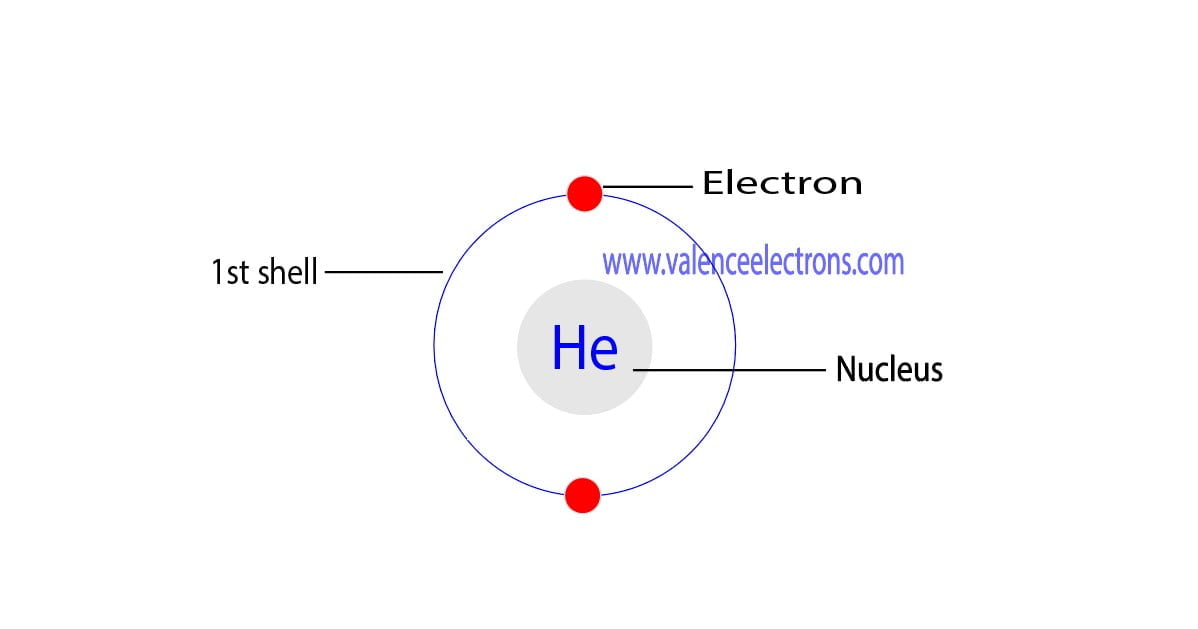
The atomic number is the number of electrons in that element. The atomic number of helium is 2. That is, the number of electrons in helium is 2. Therefore, a helium atom will have two electrons in the first shell.
The Bohr atomic model has many limitations. In the Bohr atomic model, the electrons can only be arranged in different shells but the exact position, orbital shape, and spin of the electron cannot be determined.
Also, electrons can be arranged correctly from 1 to 18 elements. The electron arrangement of any element with atomic number greater than 18 cannot be accurately determined by the Bohr atomic model following the 2n2 formula. We can overcome all limitations of the Bohr model following the electron configuration through orbital.
Electron configuration of helium through orbital
Atomic energy shells are subdivided into sub-energy levels. These sub-energy levels are also called orbital. The most probable region of electron rotation around the nucleus is called the orbital.
The sub-energy levels depend on the azimuthal quantum number. It is expressed by ‘l’. The value of ‘l’ is from 0 to (n – 1). The sub-energy levels are known as s, p, d, and f.
| Orbit Number | Value of ‘l’ | Number of subshells | Number of orbitals | Subshell name | Electrons holding capacity | Electron configuration |
| 1 | 0 | 1 | 1 | 1s | 2 | 1s2 |
| 2 | 0 1 | 2 | 1 3 | 2s 2p | 2 6 | 2s2 2p6 |
| 3 | 0 1 2 | 3 | 1 3 5 | 3s 3p 3d | 2 6 10 | 3s2 3p6 3d10 |
| 4 | 0 1 2 3 | 4 | 1 3 5 7 | 4s 4p 4d 4f | 2 6 10 14 | 4s2 4p6 4d10 4f14 |
Explanation:
- If n = 1,
(n – 1) = (1–1) = 0
Therefore, the value of ‘l’ is 0. So, the sub-energy level is 1s. - If n = 2,
(n – 1) = (2–1) = 1.
Therefore, the value of ‘l’ is 0, 1. So, the sub-energy levels are 2s, and 2p. - If n = 3,
(n – 1) = (3–1) = 2.
Therefore, the value of ‘l’ is 0, 1, 2. So, the sub-energy levels are 3s, 3p, and 3d. - If n = 4,
(n – 1) = (4–1) = 3
Therefore, the value of ‘l’ is 0, 1, 2, 3. So, the sub-energy levels are 4s, 4p, 4d, and 4f. - If n = 5,
(n – 1) = (n – 5) = 4.
Therefore, l = 0,1,2,3,4. The number of sub-shells will be 5 but 4s, 4p, 4d, and 4f in these four subshells it is possible to arrange the electrons of all the elements of the periodic table.
| Sub-shell name | Name source | Value of ‘l’ | Value of ‘m’ (0 to ± l) | Number of orbital (2l+1) | Electrons holding capacity 2(2l+1) |
| s | Sharp | 0 | 0 | 1 | 2 |
| p | Principal | 1 | −1, 0, +1 | 3 | 6 |
| d | Diffuse | 2 | −2, −1, 0, +1, +2 | 5 | 10 |
| f | Fundamental | 3 | −3, −2, −1, 0, +1, +2, +3 | 7 | 14 |
The orbital number of the s-subshell is one, three in the p-subshell, five in the d-subshell, and seven in the f-subshell. Each orbital can have a maximum of two electrons.
The sub-energy level ‘s’ can hold a maximum of two electrons, ‘p’ can hold a maximum of six electrons, ‘d’ can hold a maximum of ten electrons, and ‘f’ can hold a maximum of fourteen electrons.
Aufbau is a German word, which means building up. The main proponents of this principle are scientists Niels Bohr and Pauli. The Aufbau method is to do electron configuration through the sub-energy level.
The Aufbau principle is that the electrons present in the atom will first complete the lowest energy orbital and then gradually continue to complete the higher energy orbital.
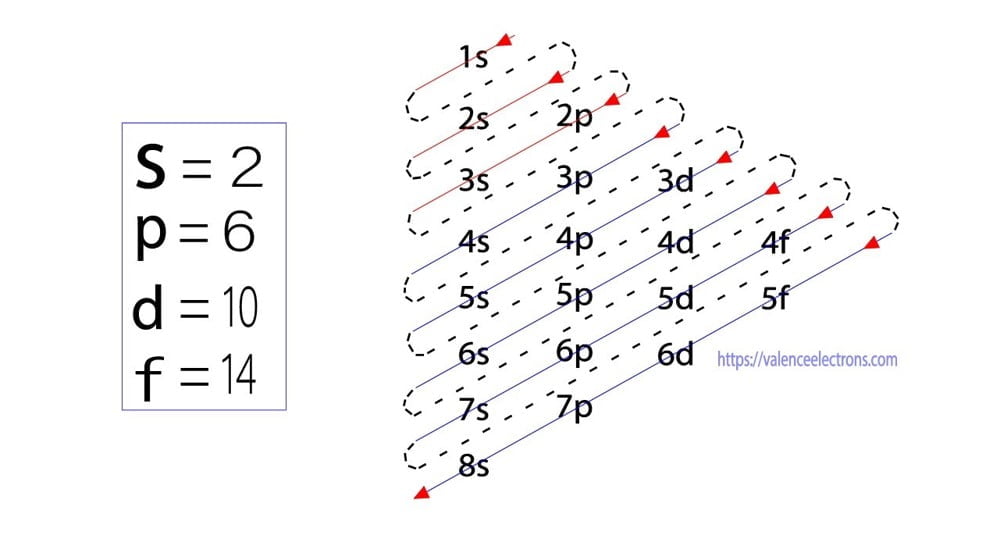
The energy of an orbital is calculated from the value of the principal quantum number ‘n’ and the azimuthal quantum number ‘l’. The orbital for which the value of (n + l) is lower is the low energy orbital and the electron will enter that orbital first.
| Orbital | Orbit (n) | Azimuthal quantum number (l) | Orbital energy (n + l) |
| 3d | 3 | 2 | 5 |
| 4s | 4 | 0 | 4 |
Here, the energy of 4s orbital is less than that of 3d. So, the electron will enter the 4s orbital first and enter the 3d orbital when the 4s orbital is full. Following the Aufbau principle, the sequence of entry of electrons into orbitals is 1s 2s 2p 3s 3p 4s 3d 4p 5s 4d 5p 6s 4f 5d 6p 7s 5f 6d 7p.

Therefore, the complete electron configuration for helium should be written as 1s2.
How to draw the orbital diagram of helium
The orbital diagram of helium is a graphical representation of the electron configuration of helium atom. This diagram shows how the electrons in the helium atom are arranged in different orbitals and indicates the spin of electrons. Orbital is the region of space around the nucleus of an atom where electrons are found.
To draw an orbital diagram of any atom, you first need to know the atomic orbitals and the orbital notation for that atom, which is already discussed in above. Also you need to know the Hund’s and Paul exclusion principle. Two fundamental principles, Hund’s Rule and the Pauli Exclusion Principle, explain how these electrons are arranged in helium’s orbital, contributing to its stability as a noble gas. Below, we define each principle and apply it to helium.
Hund’s Rule
Hund’s Rule states that when electrons fill orbitals of the same energy (called degenerate orbitals, like the three 2p orbitals), they spread out to occupy each orbital singly before pairing up. Additionally, these single electrons have the same spin (e.g., all spin-up, ↑) to minimize electron-electron repulsion and maximize stability.
Pauli Exclusion Principle
The Pauli Exclusion Principle states that no two electrons in an atom can have the same set of four quantum numbers. In practice, this means each orbital can hold a maximum of two electrons, and these electrons must have opposite spins (e.g., one ↑ and one ↓).
Orbital diagrams are usually represented by boxes. Each box represents an orbital and the arrows within the box represent the position of the electron. The boxes are arranged in order of energy of the orbitals. The lowest energy orbitals are closest to the nucleus and the higher energy orbitals are progressively further away from the nucleus in order of their energy levels.
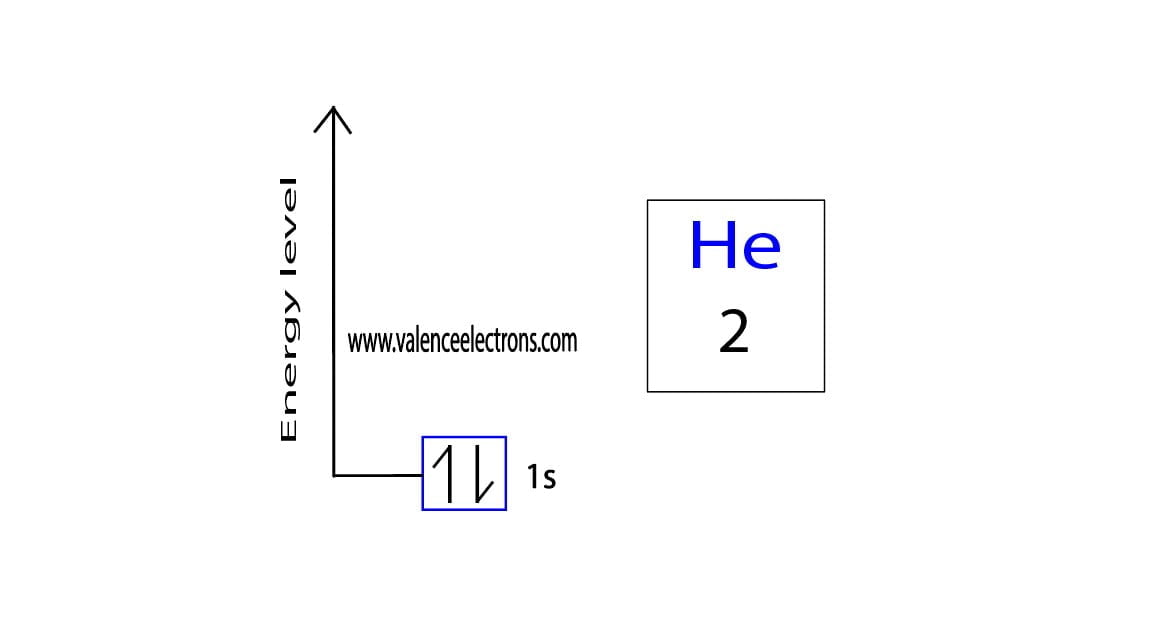
To write the orbital diagram of helium, the 1s orbital is the closest and lowest energy orbital to the nucleus. Therefore, the electrons will first enter the 1s orbital. According to Hund’s principle, the first electron will enter the 1s orbital in the clockwise direction and the remaining one will enter the 1s orbital in the anti-clockwise direction. This is clearly shown in the figure of the orbital diagram of helium.
Why This Matters for Helium
Determine the group and period of helium through the electron configuration
The electron configuration shows that the helium atom has only an orbit(shell). The last orbit is the period of that element. That is, the period of helium is 1.
Again, helium is an inert element. Therefore, helium is placed in group 18 with all the inert elements of the periodic table. Therefore, we can say that the periodic table of helium is located in period 1 and group 18.
Determine the valency and valence electrons of helium
The ability of an atom of an element to combine with another atom during the formation of a compound is called valency. Helium is an inert element. Therefore, the valency of helium is zero.
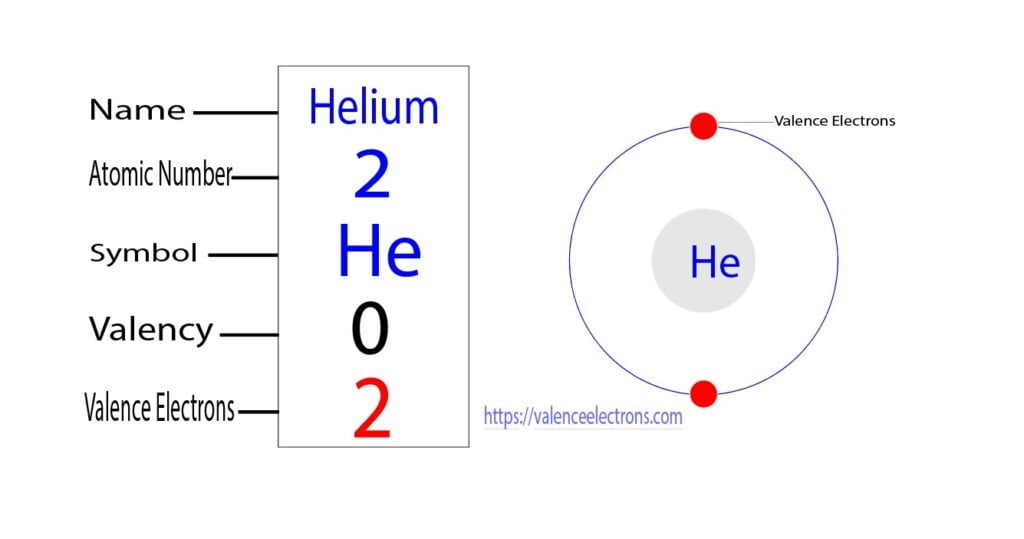
On the other hand, the number of electrons in the last orbit of an element is the valence electrons of that element. The last orbit of helium has two electrons. Therefore, the valence electrons of helium are two.
Reasons for placing helium in group 18
The electron configuration of helium shows that the number of electrons in the last orbit of the helium atom is two. We know that the number of electrons in the last orbit of an element is the number of groups in that element.
Accordingly, the group of helium is two but helium is an inert element. All inert elements are placed in group number 18 in the periodic table. Therefore, helium is placed in group 18 instead of group 2.
Why is helium an inert gas?
The elements in group 18 of the periodic table are inert gases. The inert gases of Group-18 are helium (He), neon(Ne), argon(Ar), krypton(Kr), xenon(Xe), and radon(Rn).
We know that the element in group 18 is helium(He). The electron configuration shows that the orbit at the end of helium is filled with electrons. Helium does not want to exchange or share any electrons because the last orbit of helium is full of electrons.
And helium does not form any compounds because it does not share any electrons. They do not participate in chemical bonding and chemical reactions. For this, they are called inert elements. The inert elements are in the form of gases at normal temperatures. For this inert elements are called inert gases.
Electron addiction to helium
The energy emitted to convert one mole of electrons into a negative ion by inserting one mole of electrons into a gaseous atom of an element in a gaseous state is called the electron addiction of that element. Electron addiction has a periodicity.
Moving from left to right of the same period in the periodic table increases the atomic radius of the elements and decreases the value of electron addiction. The helium atoms are located on the right side of the periodic table.
And helium inert gas. For this, the helium atom has no electron addiction. On the other hand, The electron configuration for helium shows that the last orbital(1s) electron of helium is full. The tendency of the element to receive electrons is the electron addiction of the element.
Helium does not exchange any electrons. This is because the last orbital of the helium is full of electrons. There is no electron addiction in helium for not accepting or rejecting electrons. For this, the electron addiction value of helium is 0.
Electronegativity of helium
Helium is an inert gas. Elements that have ionic energy and electron addiction also have electronegativity. It has no electron addiction. Therefore, Helium does not have electronegativity. The value of helium’s electronegativity is 0.
Ionic properties of the helium atom
Helium is an electron-filled element. The last orbit of helium is full of electrons. Therefore, helium is an inert element and helium atoms are more stable.
Therefore, helium atoms do not want to become ions very easily. It takes a lot of energy to turn a helium atom into an ion. The ionization energy of helium is 2372.3 kJ/mol and 5250.5 kJ/mol.
Characteristics of the helium atom
- The atomic number of helium is 2. The atomic number of an element is the number of electrons in that element. Therefore, the number of electrons in helium is two.
- Helium is an s-block element.
- Helium is an inert element.
- The valency of helium is 0 and the valence electrons are two.
- Period-1 and group-18.
- The standard atomic weight of helium is 4.002602.
- The melting point is 0.95 K (−272.20 ° C, −457.96 ° F) (at 2.5 MPa) and the boiling point is 4.222 K (−268.928 C, −452.070 ° F).
- Helium is in the form of gas at normal temperature.
- Helium has no electron addiction. That is, the electron addiction of helium is 0.
- Helium has no electronegativity.
- There is no helium oxidation number.
- Helium is an inert gas. Therefore, helium does not form any bonds or compounds.
- Helium does not form bonds with itself and does not form bonds with other elements.
- The ionization energy of helium is 2372.3 kJ/mol and 5250.5 kJ/mol.
Reference
- Fursa, D. V., & Bray, I. (1995). Calculation of electron-helium scattering. Physical Review A, 52(2), 1279.
- Slater, J. C. (1928). The normal state of helium. Physical Review, 32(3), 349.
- Rosenblit, M., & Jortner, J. (2006). Electron bubbles in helium clusters. I. Structure and energetics. The Journal of chemical physics, 124(19).
This post on Helium’s electron configuration and orbital diagram is incredibly informative! I appreciate the clear explanations and visuals that make complex concepts easier to understand. It’s fascinating to see how Helium fits into the broader context of the periodic table. Thank you for sharing such valuable insights!
Great explanation of helium’s electron configuration! I loved how you broke down the orbital diagram—it really helped clarify the concept for me. Thanks for making it so accessible!