Krypton Electron Configuration: [Ar] 3d¹⁰ 4s² 4p⁶ Explained
The electron configuration of krypton is 1s2 2s2 2p6 3s2 3p6 4s2 3d10 4p6 which means that the first two electrons enter the 1s orbital. Since the 1s orbital can hold only two electrons, the next two will enter the 2s orbital. The next six electrons enter the 2p subshell. The p subshell can hold a maximum of six electrons. So first we put six electrons in the 2p subshell and then the next two electrons in the 3s orbital.
Since the 3s is now full, the electrons will move to the 3p subshell, where the next six electrons will enter. The 3p subshell is now full. Consequently, the following two electrons will enter the 4s orbital. Since the 4s orbital is full, the next ten electrons will move into the 3d subshell. The 3d subshell is now full. Consequently, the remaining six electrons will enter the 4p subshell. Hence, the electron configuration of krypton will be 1s2 2s2 2p6 3s2 3p6 4s2 3d10 4p6.
Krypton is the 36th element in the periodic table and the symbol is ‘Kr’. Krypton has an atomic number of 36, which means that its atom has 36 electrons around its nucleus. The electron configuration of krypton refers to the arrangement of electrons in the krypton atom’s orbitals. It describes how electrons are distributed among the various atomic orbitals and energy levels, and provides a detailed map of where each electron is likely to be found.
To understand the mechanism of krypton electron configuration, you must understand two basic things. These are orbits and orbitals. Also, you can arrange electrons in those two ways. In this article, I have discussed all the necessary points to understand the mechanism of krypton electron configuration, including noble gas notation, orbital diagram, valence electrons, and full & abbreviated configurations. I hope this will be helpful in your study.
Electron arrangement of Krypton through Bohr Model
![Krypton Electron Configuration: [Ar] 3d¹⁰ 4s² 4p⁶ Explained 2 Krypton atom electron configuration](https://valenceelectrons.com/wp-content/uploads/2022/01/Krypton-Kr-atom-electron-configuration-Bohr-model.jpg)
Scientist Niels Bohr was the first to give an idea of the atom’s orbit. He provided a model of the atom in 1913 and provided a complete idea of orbit in that model.
The electrons of the atom revolve around the nucleus in a certain circular path. These circular paths are called orbits (shells or energy levels). These orbits are expressed by n. [n = 1,2,3,4 . . . The serial number of the orbit]
The name of the first orbit is K, L is the second, M is the third, and N is the name of the fourth orbit. The electron holding capacity of each orbit is 2n2.
| Shell Number (n) | Shell Name | Electrons Holding Capacity (2n2) |
| 1 | K | 2 |
| 2 | L | 8 |
| 3 | M | 18 |
| 4 | N | 32 |
Explanation:
- Let, n = 1 for K orbit. So, the maximum electron holding capacity in the K orbit is 2n2 = 2 × 12 = 2 electrons.
- n = 2, for L orbit. The maximum electron holding capacity in the L orbit is 2n2 = 2 × 22 = 8 electrons.
- n=3 for M orbit. The maximum electron holding capacity in the M orbit is 2n2 = 2 × 32 = 18 electrons.
- n=4 for N orbit. The maximum electron holding capacity in N orbit is 2n2 = 2 × 42 = 32 electrons.
Therefore, the maximum electron holding capacity in the first shell is two, the second shell is eight and the 3rd shell can have a maximum of eighteen electrons.
![Krypton Electron Configuration: [Ar] 3d¹⁰ 4s² 4p⁶ Explained 3 Position of krypton(Kr) in the periodic table](https://valenceelectrons.com/wp-content/uploads/2021/11/Position-of-kryptonKr-in-the-periodic-table-1024x375.jpg)
The atomic number is the number of electrons in that element. The atomic number of krypton is 36. That is, the number of electrons in krypton is thirty-six. Therefore, the krypton atom will have two electrons in the first shell, eight in the 2nd orbit, eighteen electrons in the 3rd shell, and the remaining eight electrons will be in the fourth shell. Therefore, the order of the number of electrons in each shell of the krypton atom is 2, 8, 18, 8.
The Bohr atomic model has many limitations. In the Bohr atomic model, the electrons can only be arranged in different shells but the exact position, orbital shape, and spin of the electron cannot be determined.
Also, electrons can be arranged correctly from 1 to 18 elements. The electron arrangement of any element with atomic number greater than 18 cannot be accurately determined by the Bohr atomic model following the 2n2 formula. We can overcome all limitations of the Bohr model following the electron configuration through orbital.
Electron configuration of krypton through Aufbau Model
Atomic energy shells are subdivided into sub-energy levels. These sub-energy levels are also called orbital. The most probable region of electron rotation around the nucleus is called the orbital.
The sub-energy levels depend on the azimuthal quantum number. It is expressed by ‘l’. The value of ‘l’ is from 0 to (n – 1). The sub-energy levels are known as s, p, d, and f.
| Orbit Number | Value of ‘l’ | Number of subshells | Number of orbitals | Subshell name | Electrons holding capacity | Electron configuration |
| 1 | 0 | 1 | 1 | 1s | 2 | 1s2 |
| 2 | 0 1 | 2 | 1 3 | 2s 2p | 2 6 | 2s2 2p6 |
| 3 | 0 1 2 | 3 | 1 3 5 | 3s 3p 3d | 2 6 10 | 3s2 3p6 3d10 |
| 4 | 0 1 2 3 | 4 | 1 3 5 7 | 4s 4p 4d 4f | 2 6 10 14 | 4s2 4p6 4d10 4f14 |
Explanation:
- If n = 1,
(n – 1) = (1–1) = 0
Therefore, the value of ‘l’ is 0. So, the sub-energy level is 1s. - If n = 2,
(n – 1) = (2–1) = 1.
Therefore, the value of ‘l’ is 0, 1. So, the sub-energy levels are 2s, and 2p. - If n = 3,
(n – 1) = (3–1) = 2.
Therefore, the value of ‘l’ is 0, 1, 2. So, the sub-energy levels are 3s, 3p, and 3d. - If n = 4,
(n – 1) = (4–1) = 3
Therefore, the value of ‘l’ is 0, 1, 2, 3. So, the sub-energy levels are 4s, 4p, 4d, and 4f. - If n = 5,
(n – 1) = (n – 5) = 4.
Therefore, l = 0,1,2,3,4. The number of sub-shells will be 5 but 4s, 4p, 4d, and 4f in these four subshells it is possible to arrange the electrons of all the elements of the periodic table.
| Sub-shell name | Name source | Value of ‘l’ | Value of ‘m’ (0 to ± l) | Number of orbital (2l+1) | Electrons holding capacity 2(2l+1) |
| s | Sharp | 0 | 0 | 1 | 2 |
| p | Principal | 1 | −1, 0, +1 | 3 | 6 |
| d | Diffuse | 2 | −2, −1, 0, +1, +2 | 5 | 10 |
| f | Fundamental | 3 | −3, −2, −1, 0, +1, +2, +3 | 7 | 14 |
The orbital number of the s-subshell is one, three in the p-subshell, five in the d-subshell, and seven in the f-subshell. Each orbital can have a maximum of two electrons.
The sub-energy level ‘s’ can hold a maximum of two electrons, ‘p’ can hold a maximum of six electrons, ‘d’ can hold a maximum of ten electrons, and ‘f’ can hold a maximum of fourteen electrons.
Aufbau is a German word, which means building up. The main proponents of this principle are scientists Niels Bohr and Pauli. The Aufbau method is to do electron configuration through the sub-energy level.
The Aufbau principle is that the electrons present in the atom will first complete the lowest energy orbital and then gradually continue to complete the higher energy orbital.
![Krypton Electron Configuration: [Ar] 3d¹⁰ 4s² 4p⁶ Explained 4 Electron configuration Mechanism Through Aufbau principal](https://valenceelectrons.com/wp-content/uploads/2021/12/Electron-configuration-via-Aufbaw-principal.jpg)
The energy of an orbital is calculated from the value of the principal quantum number ‘n’ and the azimuthal quantum number ‘l’. The orbital for which the value of (n + l) is lower is the low energy orbital and the electron will enter that orbital first.
| Orbital | Orbit (n) | Azimuthal quantum number (l) | Orbital energy (n + l) |
| 3d | 3 | 2 | 5 |
| 4s | 4 | 0 | 4 |
Here, the energy of 4s orbital is less than that of 3d. So, the electron will enter the 4s orbital first and enter the 3d orbital when the 4s orbital is full. Following the Aufbau principle, the sequence of entry of electrons into orbitals is 1s 2s 2p 3s 3p 4s 3d 4p 5s 4d 5p 6s 4f 5d 6p 7s 5f 6d 7p.
Therefore, the complete electron configuration for krypton should be written as 1s2 2s2 2p6 3s2 3p6 4s2 3d10 4p6.
![Krypton Electron Configuration: [Ar] 3d¹⁰ 4s² 4p⁶ Explained 5 krypton electron configuration](https://valenceelectrons.com/wp-content/uploads/2021/11/KryptonKr-electron-configuration-1024x460.jpg)
Note: The unabbreviated electron configuration of krypton is [Ar] 3d10 4s2 4p6. When writing an electron configuration, you have to write serially.
How to write the orbital diagram for Krypton
Orbital diagrams are usually represented by boxes. Each box represents an orbital and the arrows within the box represent the position of the electron. The boxes are arranged in order of energy of the orbitals.
The lowest energy orbitals are closest to the nucleus and the higher energy orbitals are progressively further away from the nucleus in order of their energy levels. To write the orbital diagram of krypton, you have to write the orbital notation of krypton. Which has been discussed in detail above.
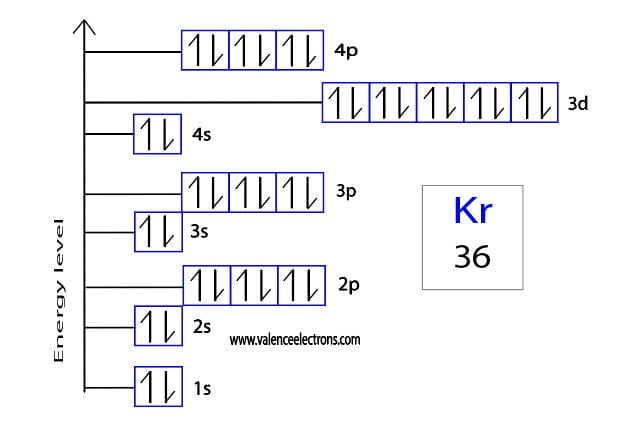
1s is the closest and lowest energy orbital to the nucleus. Therefore, the electrons will first enter the 1s orbital. According to Hund’s principle, the first electron will enter 1s orbital in the clockwise direction and the next electron will enter the 1s orbital in the anti-clockwise direction.
The 1s orbital is now filled with two electrons. The next two electrons will enter the 2s orbital just like the 1s orbital. The next three electrons will enter the 2p orbital in the clockwise direction and the next three electrons will enter the 2p orbital in the anti-clockwise direction.
The 2p orbital is now full. Then the next two electrons will enter the 3s orbital just like the 1s orbital. Then the next three electrons will enter the 3p orbital in the clockwise direction and the next three electrons will enter the 3p orbital in the anti-clockwise direction.
The 3p orbital is now full. So, the next two electrons will enter the 4s orbital just like the 1s orbital. The 4s orbital is now full. Therefore, the next five electrons will enter the 3d orbital in the clockwise direction and the next five electrons will enter the 3d orbital in the anti-clockwise direction.
The 3d orbital is now full. So, the next three electrons will enter the 4p orbital in the clockwise direction and the remaining three electrons will enter the 4p orbital in the anti-clockwise direction. This is clearly shown in the figure of the orbital diagram of krypton.
Try the Orbital Diagram Calculator and get instant results for any element.
Why is krypton inert gas?
Ground state electron configuration of krypton is 1s2 2s2 2p6 3s2 3p6 3d10 4s2 4p6. This electron configuration shows that the last shell of krypton has eight electrons. Therefore, the valence electrons of krypton are eight.
The elements in group-18 of the periodic table are inert gases. The inert gases of Group-18 are helium(He), neon(Ne), argon(Ar), krypton(Kr), xenon(Xe), and radon(Rn).
We know that the element in group-18 is krypton. The electron configuration shows that the orbit at the end of krypton is filled with electrons. Krypton does not want to exchange or share any electrons because the last orbit is full of electrons and krypton does not form any compounds because it does not share any electrons.
They do not participate in chemical bonding and chemical reactions. For this, they are called inert elements. The inert elements are in the form of gases at normal temperatures. For this inert elements are called inert gases. Again for this same reason, inert gas is called a noble gas.
![Krypton Electron Configuration: [Ar] 3d¹⁰ 4s² 4p⁶ Explained 1 Electron Configuration Calculator](https://valenceelectrons.com/wp-content/uploads/2023/03/Electron-Configuration-Calculator.jpg)
Great explanation of krypton’s electron configuration! I appreciate how you broke down the notation and provided context for each subshell. It makes understanding the arrangement so much easier for someone just starting to learn about electron configurations.
Great explanation of Krypton’s electron configuration! It’s fascinating how the structure impacts its properties. I appreciate the breakdown of each subshell—makes it much easier to understand!
Great explanation of Krypton’s electron configuration! I appreciate how you broke down the notation and related it to the previous noble gas. It really helped clarify the concept for me.
This explanation of Krypton’s electron configuration is really helpful! I appreciate how you broke down the notation and clarified the significance of each subshell. It makes understanding the element’s chemistry so much easier. Thank you for the insights!