How to Find the Valence Electrons for ClF3 (Chlorine Trifluoride)?
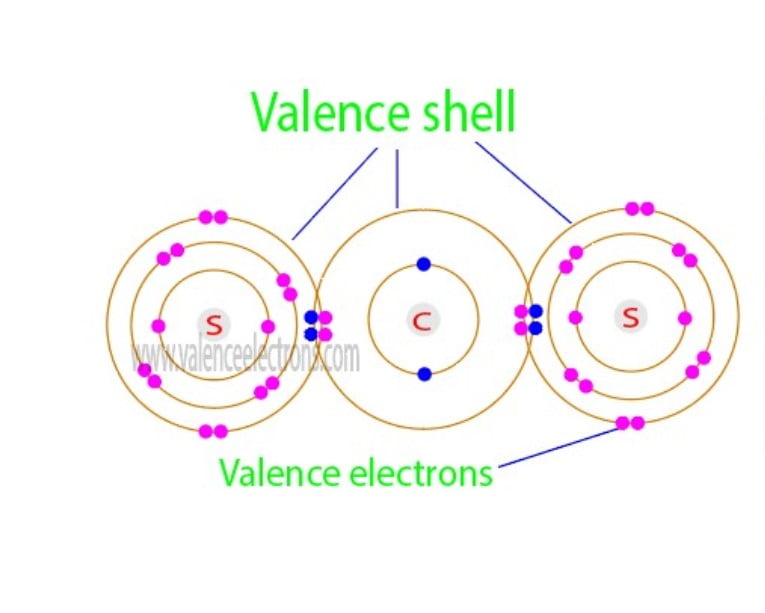
The total number of electrons in the last orbit of an element is called the valence electrons of that element. Two or more elements combine to form compounds.
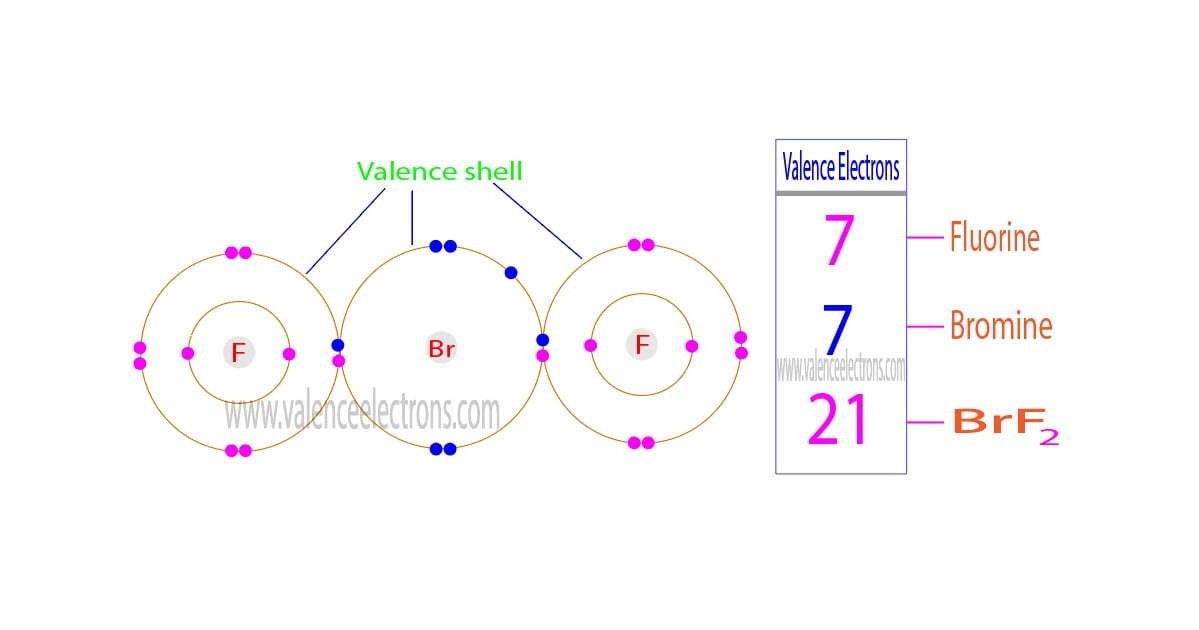
The valence electrons of a compound are the sum of the total valence electrons of each element in that compound. Chlorine trifluoride consists of two basic atoms. One is a chlorine atom and the other is a fluorine atom.
The valence electrons of chlorine trifluoride are the sum of the total valence electrons of chlorine and fluorine in the compound ClF3. The chlorine trifluoride compound has a total of twenty-eight electrons in the last orbits of chlorine and fluorine.
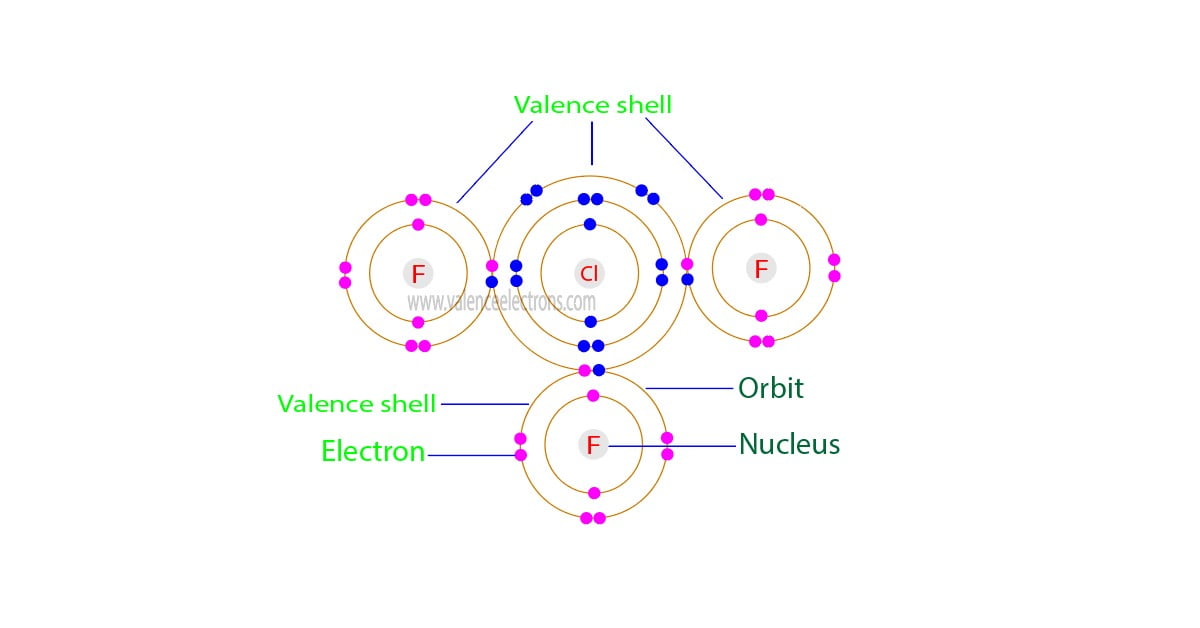
Hence, the total number of valence electrons in chlorine trifluoride is twenty-eight. To determine the valence electrons of a compound, the number of valence electrons in each element of the compound must be known.
This article discusses in detail how to determine the valence electrons of chlorine trifluoride very easily. Hopefully, after reading this article you will know more about this topic.
How to easily determine the number of valence electrons of chlorine trifluoride?
The symbol form of chlorine trifluoride is ClF3. To determine the valence electrons of ClF3, it is first necessary to know the valence electrons of the fluorine and chlorine atoms. To determine the valence electrons of chlorine trifluoride we have to follow two steps. It is shown below:
Step 1: Determine the valence electrons of chlorine and fluorine atoms
The atomic number of chlorine is 17. So its total number of electrons is seventeen. The electron configuration of chlorine shows that it has seven electrons in its last orbit.
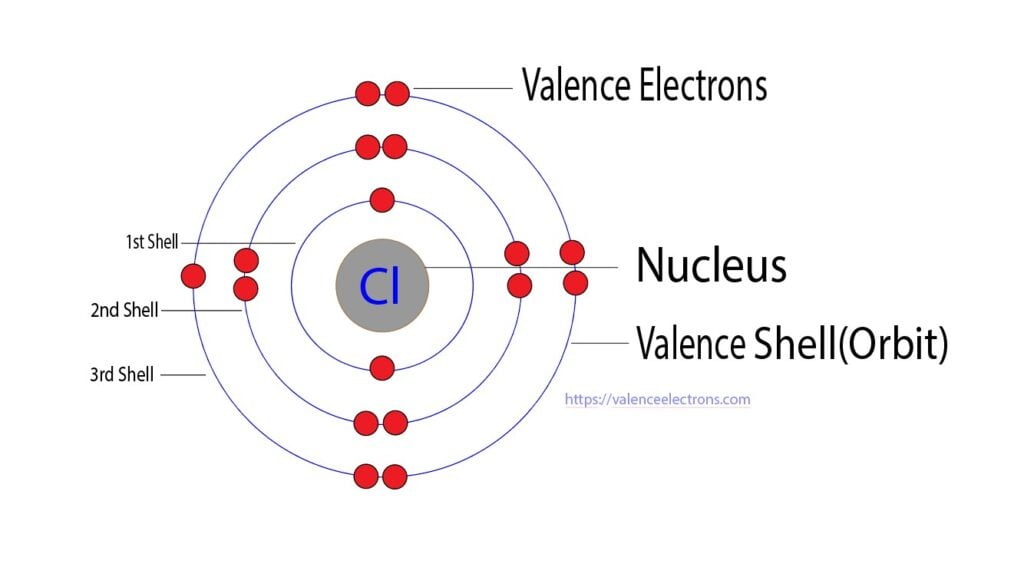
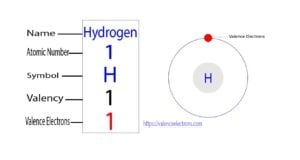
We already know that the electrons in the last orbit of an element are the valence electrons of that element. Therefore, the valence electrons of chlorine are seven.
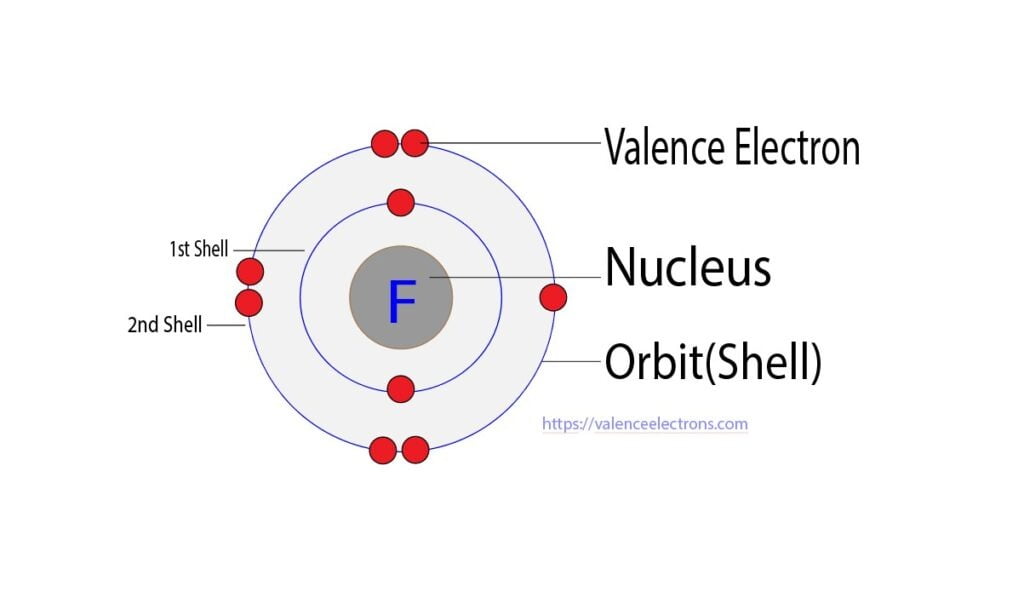
On the other hand, the atomic number of fluorine is 9. So its total number of electrons is nine. The electron configuration of fluorine shows that it has seven electrons in its last orbit. Therefore, the valence electrons of fluorine are seven.
Step 2: Determine the total number of valence electrons in the chlorine trifluoride compound
Chlorine trifluoride is a compound. It is composed of one chlorine atom and three fluorine atoms. Therefore, adding the valence electrons of one chlorine atom and the valence electrons of three fluorine atoms can easily determine the valence electrons of chlorine trifluoride.
Mathematical Analysis:
ClF3
= 7 + (7×3)
= 28
| Chlorine (Cl) | Fluorine (F) | Chlorine trifluoride (ClF3) |
| 7 | 7 | 28 |
From the above mathematical analysis, we can see that the total number of electrons in the outermost orbit of chlorine trifluoride is twenty-eight. Therefore, the valence electrons of chlorine trifluoride are twenty-eight.
Why do you need to know the valence electrons of an element?
In addition to knowing the number of electrons of an element, it is also necessary to know the number of valence electrons of that element. The number of valence electrons of an element carries important properties of that element.
Many important properties of elements and compounds can be determined by valence electrons. Some important features are mentioned below:
- Determining the element’s position in the periodic table
- Determining the number of groups of elements in the periodic table
- Determining Valency
- Lewis dot formation
- Determination of the molecular structure of the compound
- Determination of bond pairs and non-pair electrons of compounds
Valence electrons are related to the characteristics of a compound. So we need to know the valence electrons of all the elements to determine the properties of compounds.