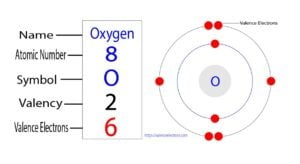
How to Find the Valence Electrons for Osmium(Os)?
The 76th element in the periodic table is osmium. The element of group-8 is osmium and its symbol is ‘Os’. Osmium is a d-block element. Therefore, the valence electrons of osmium are determined differently.
What are the valence electrons of osmium?
The valence electrons are the total number of electrons in the last orbit. The 3rd element in group-8 is osmium and it is the d-block element. The elements in groups 3-12 are called transition elements.
But in the case of transition elements, the valence electrons remain in the inner shell(orbit). Because the electron configuration of the transition elements shows that the last electrons enter the d-orbital.
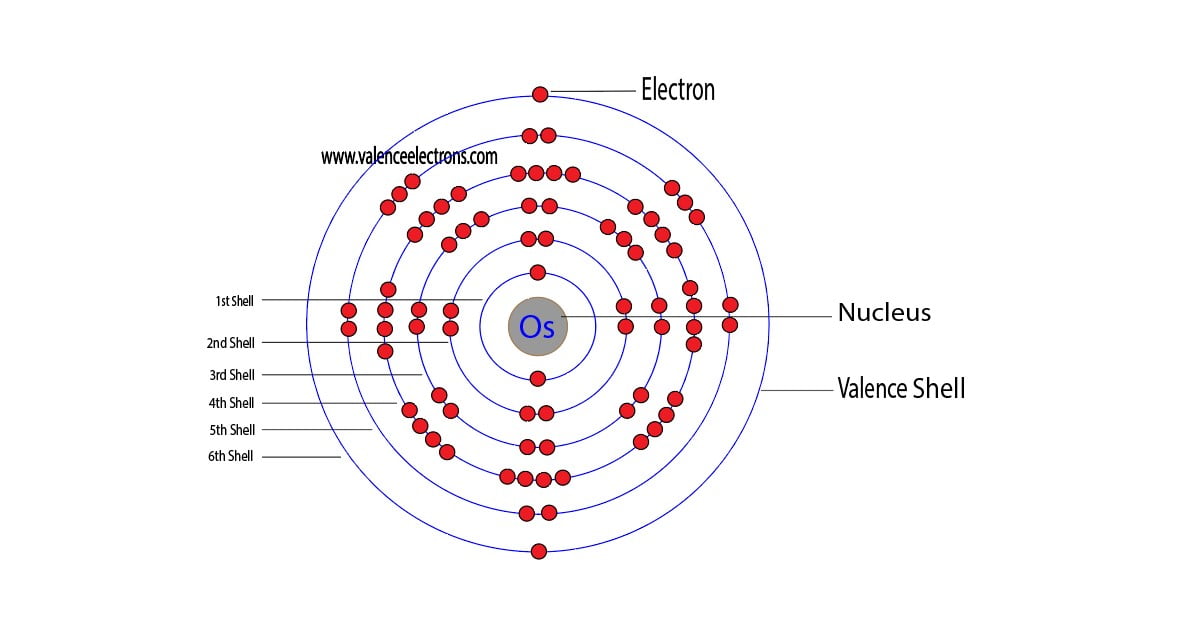
The electron configuration of osmium shows that the last shell of osmium has two electrons and the d-orbital has a total of six electrons. Therefore, the valence electrons of osmium are eight.
The valence electrons determine the properties of the element and participate in the formation of bonds. The electrons of the d-orbital participate in the formation of bonds.
So, to determine the valence electrons of a transition element, the last shell electrons have to be calculated with the d-orbital electrons.
How do you calculate the number of valence electrons in an osmium atom?
The valence electrons have to be determined by following a few steps. The electron configuration is one of them. It is not possible to determine the valence electrons without electron configuration.
Knowing the electron configuration in the right way, it is very easy to determine the valence electrons of all the elements.
The valence electrons of the transition element cannot be determined according to Bohr’s atomic model. Because the valence electrons of the transition elements are located in the inner shell.
However, the valence electron of the transition element can be easily determined according to the Aufbau principle. Now we will learn how to determine the valence electron of osmium.
Step-1: Determining the total number of electrons in osmium
The elements in the periodic table are arranged according to their atomic number. Since osmium is the 76th element of the periodic table, the atomic number of osmium is 76.
We must always remember that the atomic number and the number of protons of an element are equal. Therefore, an osmium atom contains seventy-six protons.
We must also remember that the number of protons and electrons in an element is equal. Therefore, an osmium atom contains seventy-six electrons in its orbits.
Step-2: Need to do electron configuration of osmium
Step 2 is very important. In this step, the electrons of osmium have to be arranged. We know that osmium atoms have a total of seventy-six electrons.
The full electron configuration of osmium is 1s2 2s2 2p6 3s2 3p6 3d10 4s2 4p6 4d10 4f14 5s2 5p6 5d6 6s2. Here the valence electron configuration of osmium is [Xe] 6s24f145d6.
Step-3: Determine the valence shell and calculate the total electrons
The third step is to diagnose the valence shell. The last shell after the electron configuration is called the valence shell. The total number of electrons in a valence shell is called valence electrons.
But the valence electrons of the transition elements are located in the inner orbit. For the transition element, the valence electrons have to be determined by adding the total electrons of the d-orbital to the electron in the last shell of the atom.
The last shell of osmium has two electrons and the d-orbital has a total of six electrons. Therefore, the valence electrons of osmium are eight.