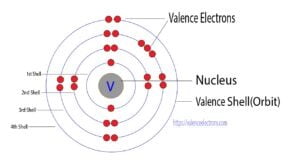
How many shells does lithium have?
Answer: Lithium has 2 electron shells.
Explanation: The electron shells, also known as energy levels, are regions around an atomic nucleus where electrons are found. Lithium (Li), with an atomic number of 3, has an electron configuration of 1s2 2s1. In this configuration, the first shell (1s) is fully occupied with 2 electrons, and the second shell (2s) contains 1 electron. Read More…