Electron Configuration for Arsenic (As, As3- ion)
Arsenic is the 33rd element in the periodic table and its symbol is ‘As’. In this article, I have discussed in detail how to easily write the complete electron configuration of arsenic.
What is the electron configuration of arsenic?
The total number of electrons in arsenic is thirty-three. These electrons are arranged according to specific rules in different orbitals.
The arrangement of electrons in arsenic in specific rules in different orbits and orbitals is called the electron configuration of arsenic.
The electron configuration of arsenic is [Ar] 3d10 4s2 4p3, if the electron arrangement is through orbitals. Electron configuration can be done in two ways.
- Electron configuration through orbit (Bohr principle)
- Electron configuration through orbital (Aufbau principle)
Electron configuration through orbitals follows different principles. For example Aufbau principle, Hund’s principle, and Pauli’s exclusion principle.
Arsenic atom electron configuration through orbit
Scientist Niels Bohr was the first to give an idea of the atom’s orbit. He provided a model of the atom in 1913. The complete idea of the orbit is given there.
The electrons of the atom revolve around the nucleus in a certain circular path. These circular paths are called orbit(shell). These orbits are expressed by n. [n = 1,2,3,4 . . . The serial number of the orbit]
K is the name of the first orbit, L is the second, M is the third, and N is the name of the fourth orbit. The electron holding capacity of each orbit is 2n2.
| Shell Number (n) | Shell Name | Electrons Holding Capacity (2n2) |
| 1 | K | 2 |
| 2 | L | 8 |
| 3 | M | 18 |
| 4 | N | 32 |
For example,
- n = 1 for K orbit.
The maximum electron holding capacity in K orbit is 2n2 = 2 × 12 = 2. - For L orbit, n = 2.
The maximum electron holding capacity in L orbit is 2n2 = 2 × 22 = 8. - n=3 for M orbit.
The maximum electron holding capacity in M orbit is 2n2 = 2 × 32 = 18. - n=4 for N orbit.
The maximum electron holding capacity in N orbit is 2n2 = 2 × 42 = 32.
Therefore, the maximum electron holding capacity in the first shell is two, the second shell is eight and the 3rd shell can have a maximum of eighteen electrons.
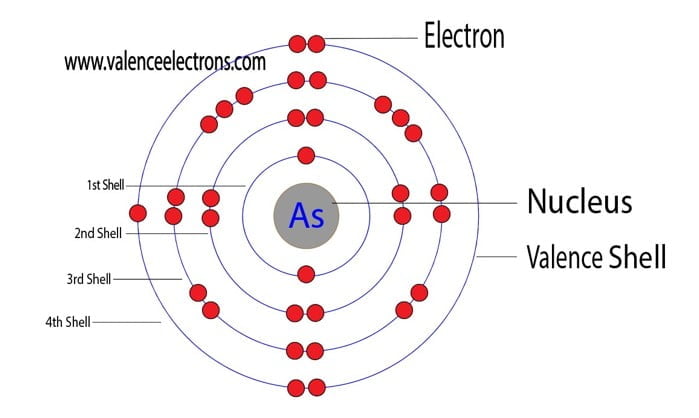
The atomic number is the number of electrons in that element. The atomic number of arsenic is 33. That is, the number of electrons in arsenic is thirty-three.
Therefore, the arsenic atom will have two electrons in the first shell, eight in the 2nd orbit, eighteen electrons in the 3rd shell, and the remaining five electrons will be in the fourth shell.
Therefore, the order of the number of electrons in each shell of the arsenic atom is 2, 8, 18, 5. Electrons can be arranged correctly through orbits from elements 1 to 18.
The electron configuration of an element with an atomic number greater than 18 cannot be properly determined according to the Bohr atomic model. The electron configuration of all the elements can be done through the orbital diagram.
Electron configuration of arsenic through orbital
Atomic energy shells are subdivided into sub-energy levels. These sub-energy levels are also called orbital. The most probable region of electron rotation around the nucleus is called the orbital.
The sub-energy levels depend on the azimuthal quantum number. It is expressed by ‘l’. The value of ‘l’ is from 0 to (n – 1). The sub-energy levels are known as s, p, d, and f.
| Orbit Number | Value of ‘l’ | Number of subshells | Number of orbital | Subshell name | Electrons holding capacity | Electron configuration |
| 1 | 0 | 1 | 1 | 1s | 2 | 1s2 |
| 2 | 0 1 | 2 | 1 3 | 2s 2p | 2 6 | 2s2 2p6 |
| 3 | 0 1 2 | 3 | 1 3 5 | 3s 3p 3d | 2 6 10 | 3s2 3p6 3d10 |
| 4 | 0 1 2 3 | 4 | 1 3 5 7 | 4s 4p 4d 4f | 2 6 10 14 | 4s2 4p6 4d10 4f14 |
For example,
- If n = 1,
(n – 1) = (1–1) = 0
Therefore, the value of ‘l’ is 0. So, the sub-energy level is 1s. - If n = 2,
(n – 1) = (2–1) = 1.
Therefore, the value of ‘l’ is 0, 1. So, the sub-energy levels are 2s, and 2p. - If n = 3,
(n – 1) = (3–1) = 2.
Therefore, the value of ‘l’ is 0, 1, 2. So, the sub-energy levels are 3s, 3p, and 3d. - If n = 4,
(n – 1) = (4–1) = 3
Therefore, the value of ‘l’ is 0, 1, 2, 3. So, the sub-energy levels are 4s, 4p, 4d, and 4f. - If n = 5,
(n – 1) = (n – 5) = 4.
Therefore, l = 0,1,2,3,4. The number of sub-shells will be 5 but 4s, 4p, 4d, and 4f in these four subshells it is possible to arrange the electrons of all the elements of the periodic table.
| Subshell name | Name source | Value of ‘l’ | Value of ‘m’ (0 to ± l) | Number of orbital (2l+1) | Electrons holding capacity 2(2l+1) |
| s | Sharp | 0 | 0 | 1 | 2 |
| p | Principal | 1 | −1, 0, +1 | 3 | 6 |
| d | Diffuse | 2 | −2, −1, 0, +1, +2 | 5 | 10 |
| f | Fundamental | 3 | −3, −2, −1, 0, +1, +2, +3 | 7 | 14 |
The orbital number of the s-subshell is one, three in the p-subshell, five in the d-subshell and seven in the f-subshell. Each orbital can have a maximum of two electrons.
The sub-energy level ‘s’ can hold a maximum of two electrons, ‘p’ can hold a maximum of six electrons, ‘d’ can hold a maximum of ten electrons, and ‘f’ can hold a maximum of fourteen electrons.

Aufbau is a German word, which means building up. The main proponents of this principle are scientists Niels Bohr and Pauli. The Aufbau method is to do electron configuration through the sub-energy level.
The Aufbau principle is that the electrons present in the atom will first complete the lowest energy orbital and then gradually continue to complete the higher energy orbital.
The energy of an orbital is calculated from the value of the principal quantum number ‘n’ and the azimuthal quantum number ‘l’. The orbital for which the value of (n + l) is lower is the low energy orbital and the electron will enter that orbital first.
| Orbital | Orbit (n) | Azimuthal quantum number (l) | Orbital energy (n + l) |
| 3d | 3 | 2 | 5 |
| 4s | 4 | 0 | 4 |
Here, the energy of 4s orbital is less than that of 3d. So, the electron will enter the 4s orbital first and enter the 3d orbital when the 4s orbital is full. The method of entering electrons into orbitals through the Aufbau principle is 1s 2s 2p 3s 3p 4s 3d 4p 5s 4d 5p 6s 4f 5d 6p 7s 5f 6d.
The first two electrons of arsenic enter the 1s orbital. The s-orbital can have a maximum of two electrons. Therefore, the next two electrons enter the 2s orbital.
The p-orbital can have a maximum of six electrons. So, the next six electrons enter the 2p orbital. The second orbit is now full. So, the remaining electrons will enter the third orbit.
Then two electrons will enter the 3s orbital of the third orbit and the next six electrons will be in the 3p orbital. The 3p orbital is now full. So, the next two electrons will enter the 4s orbital and ten electrons will enter the 3d orbital.
The 3d orbital is now full. So, the remaining three electrons enter the 4p orbital. Therefore, the arsenic full electron configuration will be 1s2 2s2 2p6 3s2 3p6 3d10 4s2 4p3.
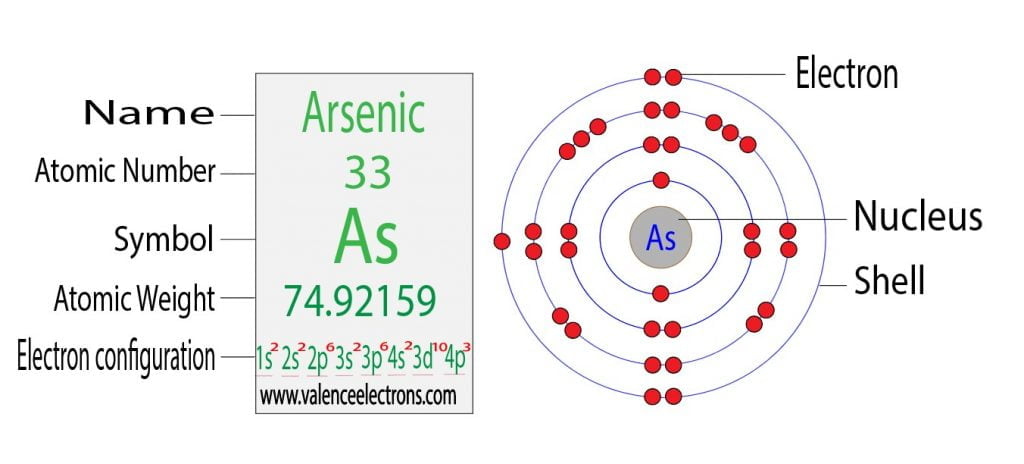
Note: The abbreviated electron configuration of arsenic is [Ar] 3d10 4s2 4p3. When writing an electron configuration, you have to write serially.
Arsenic ion(As3-) electron configuration
The ground state electron configuration of arsenic is 1s2 2s2 2p6 3s2 3p6 3d10 4s2 4p3. This electron configuration shows that the last shell of arsenic has five electrons. Therefore, the valence electrons of arsenic are five.
The elements that have 5, 6, or 7 electrons in the last shell receive the electrons in the last shell during bond formation. The elements that receive electrons and form bonds are called anions.

During the formation of a bond, the last shell of arsenic receives three electrons and turns into an arsenic ion(As3-). That is, arsenic is an anion element.
As + 3e– → As3-
The electron configuration of arsenic ion(As3-) is 1s2 2s2 2p6 3s2 3p6 4s2 3d10 4p6. This electron configuration shows that the arsenic ion(As3-) acquired the electron configuration of krypton.
Arsenic atom exhibits -3, +3, +5 oxidation state. The oxidation state of the element changes depending on the bond formation.
Video for writing the electron configuration for Arsenic (As and the As3- ion)
FAQs
What is the symbol for arsenic?
Ans: The symbol for arsenic is ‘As’.
How many electrons does arsenic have?
Ans: 33 electrons.
How do you write the complete electron configuration for arsenic?
Ans: 1s2 2s2 2p6 3s2 3p6 3d10 4s2 4p3.
What is the valency of arsenic?
Ans: The valency of arsenic is 5 and 3.