What is the Valence Electron Configuration for the Carbon Atom?
Answer: The valence electron configuration for the carbon atom is 2s2 2p2.
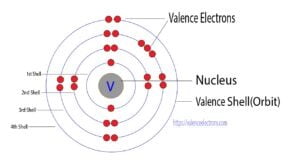
Explanation: Valence electrons are the electrons present in the outermost energy level (shell) of an atom. Carbon, with an atomic number of 6, has an electron configuration of 1s2 2s2 2p2.
In this configuration, the 2s2 and 2p2 subshells contain the valence electrons. Carbon has 4 valence electrons in total.
Significance: Carbon’s 4 valence electrons contribute to its chemical behavior and its ability to form a wide range of compounds, including organic molecules.
The arrangement of these electrons influences carbon’s reactivity and bonding patterns.