How many unpaired electrons are in the boron atom?
Answer: There is 1 unpaired electron in the boron atom.
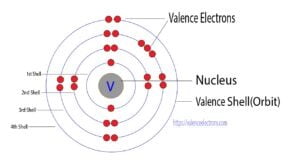
Explanation: Boron (B), with an atomic number of 5, has an electron configuration of 1s2 2s2 2p1.
In this configuration, the three electrons are distributed in the 1s, 2s, and 2p subshell.
The 2p subshell contains one unpaired electron in a single orbital (2p1).
The other two electrons (2s2) are paired in the same orbital. Therefore, boron has 1 unpaired electron.
Significance: The presence of unpaired electrons in the boron atom contributes to its chemical reactivity and its tendency to participate in chemical reactions by sharing or donating this unpaired electron.