How to Find the Valence Electrons for Fluorine (F)?
Fluorine is the 9th element in the periodic table and the first element in group-17. Fluorine is a halogen element and its symbol is ‘F’. Fluorine participates in the formation of bonds…
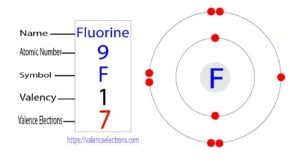
Fluorine is the 9th element in the periodic table and the first element in group-17. Fluorine is a halogen element and its symbol is ‘F’. Fluorine participates in the formation of bonds…
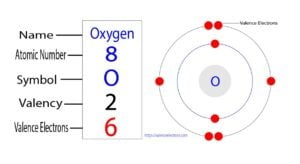
Oxygen is the eighth element of the periodic table and the symbol is ‘O’. The standard atomic mass of oxygen is 15.99903. Oxygen participates in the formation of bonds through valence electrons….
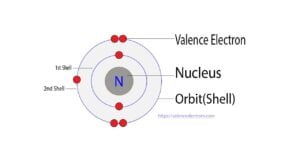
Nitrogen is the 7th element in the periodic table and the first element in group-15. The standard atomic mass of nitrogen is 14.006 and its symbol is ‘N’. Nitrogen participates in the…
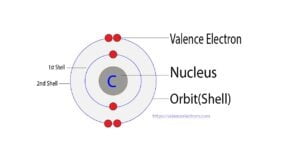
Carbon is the sixth element in the periodic table and the 1st element in group-14. The standard atomic mass of carbon is 12.0096 and its symbol is ‘C’. Carbon participates in the…
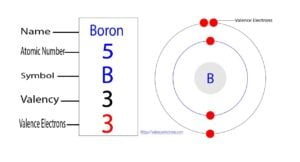
Boron is the fifth element of the periodic table and the symbol is ‘B’. Boron participates in the formation of bonds through valence electrons. This article discusses in detail how to easily…
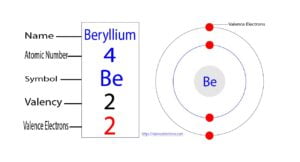
Beryllium is the fourth element of the periodic table and the symbol is ‘Be’. Beryllium participates in the formation of bonds through valence electrons. This article discusses in detail how to easily…
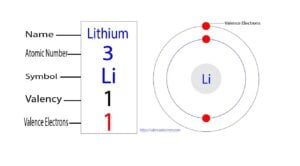
Lithium is the third element in the periodic table and the 2nd element in group-1. The standard atomic mass of lithium is 6.938 and its symbol is ‘Li’. Lithium is called alkali…
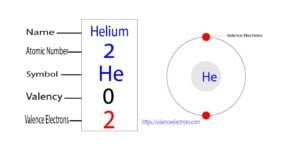
Helium is the second element in the periodic table and the first element in Group -18. The symbol for the helium element is ‘He’. Helium does not participate in chemical reactions and…
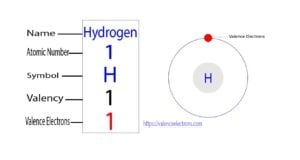
The 1st element of the periodic table is hydrogen. The element of group-1 is hydrogen and its symbol is ‘H’. Hydrogen forms bonds through its valence electrons. This article discusses in detail how…
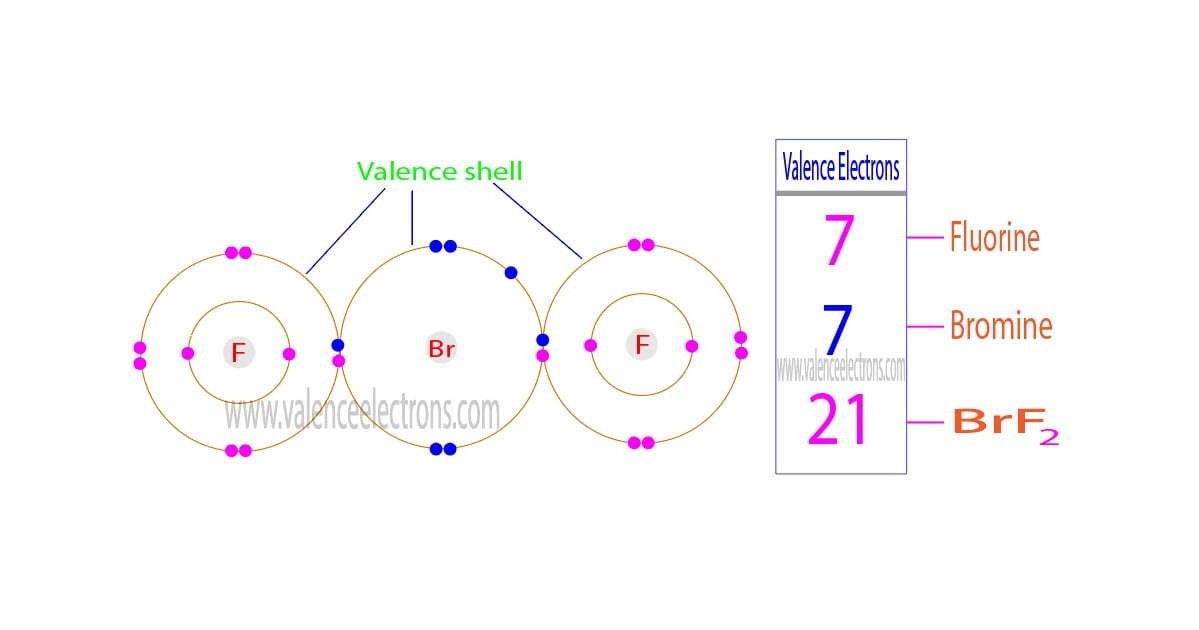
The total number of electrons in the last orbit of an element is called the valence electrons of that element. Two or more elements combine to form compounds. The valence electrons of…
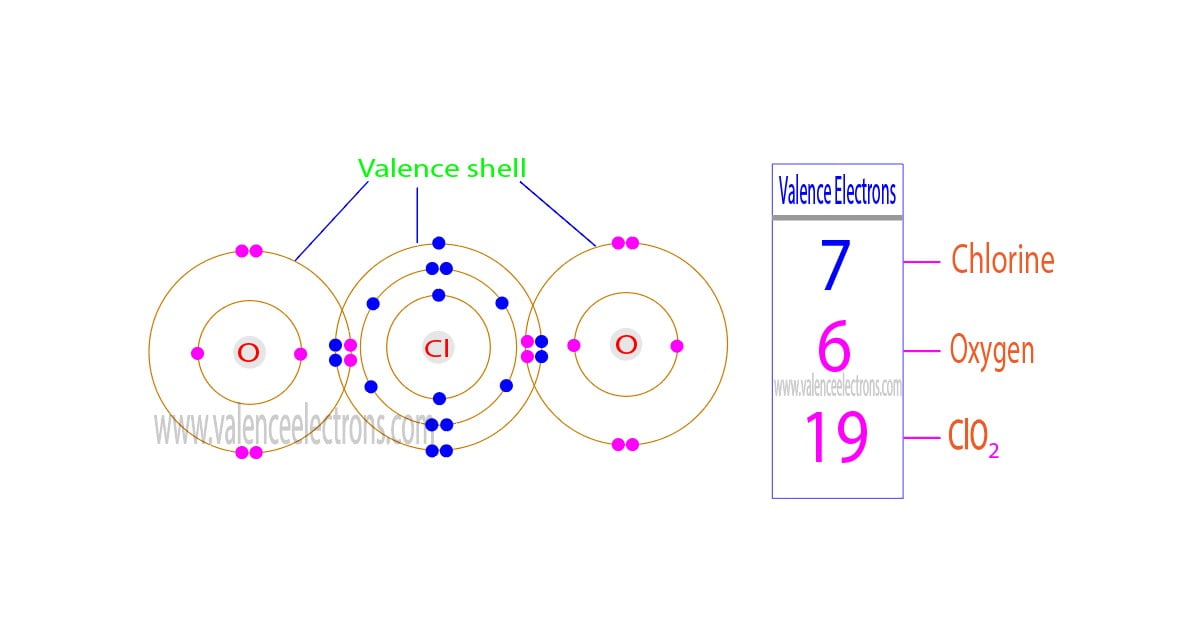
The total number of electrons in the last orbit of an element is called the valence electrons of that element. Two or more elements combine to form compounds. The valence electrons of…
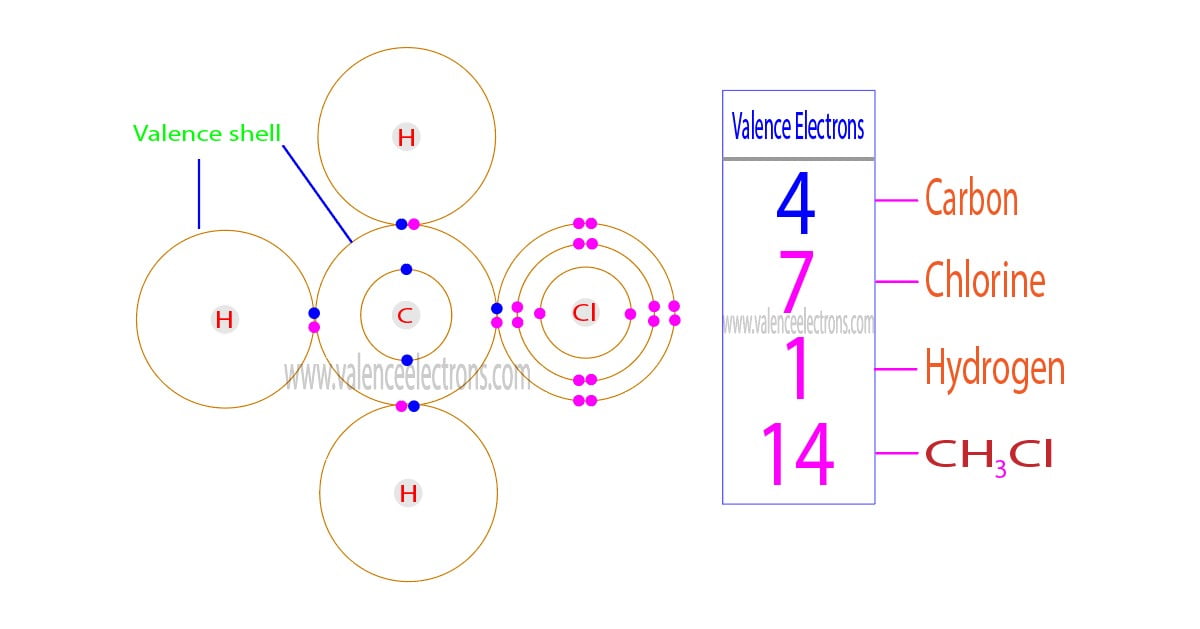
The total number of electrons in the last orbit of an element is called the valence electrons of that element. Two or more elements combine to form compounds. The valence electrons of…
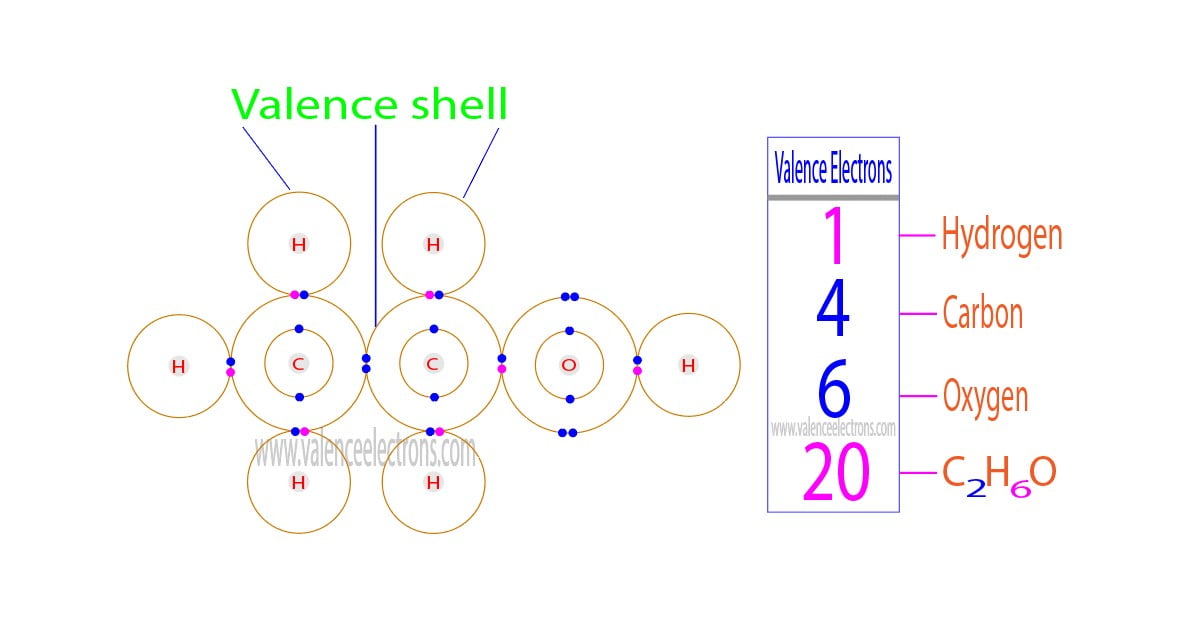
The total number of electrons in the last orbit of an element is called the valence electrons of that element. Two or more elements combine to form compounds. The valence electrons of…
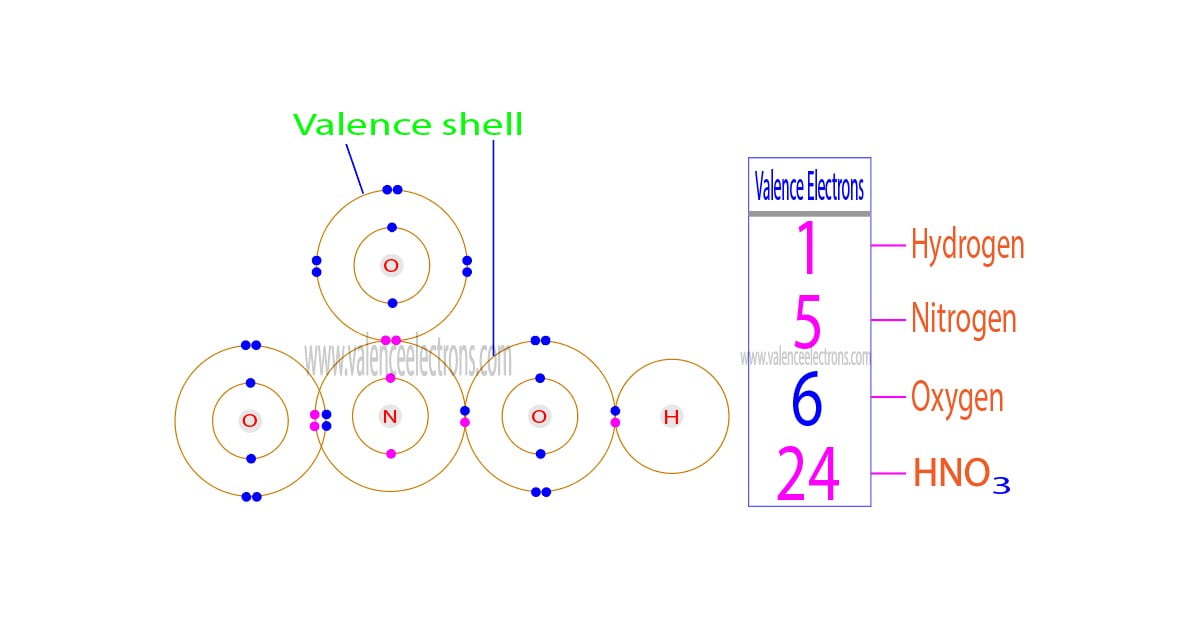
The total number of electrons in the last orbit of an element is called the valence electrons of that element. Two or more elements combine to form compounds. The valence electrons of…
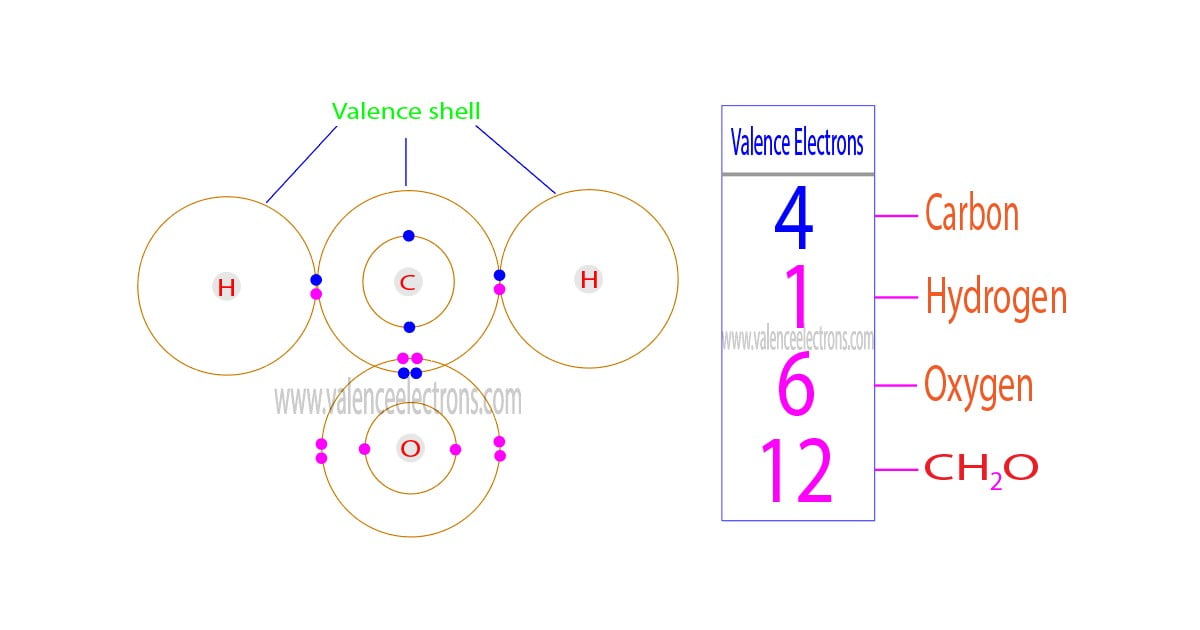
The total number of electrons in the last orbit of an element is called the valence electrons of that element. Two or more elements combine to form compounds. The valence electrons of…