How to Find the Valence Electrons for Chromium (Cr)?
The 24th element in the periodic table is chromium. The element of group-6 is chromium and its symbol is ‘Cr’. Chromium is a transition element. Therefore, the valence electrons of chromium are…
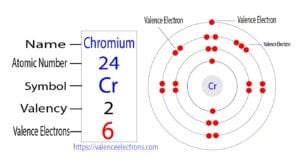
The 24th element in the periodic table is chromium. The element of group-6 is chromium and its symbol is ‘Cr’. Chromium is a transition element. Therefore, the valence electrons of chromium are…
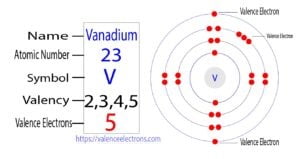
The element of group-5 is vanadium and its symbol is ‘V’. Vanadium is a transition element. Therefore, the valence electrons of vanadium are determined differently. This article discusses in detail how to…
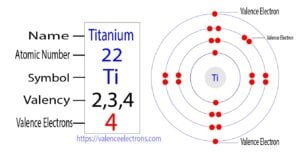
Titanium is a transition element. Therefore, the valence electrons of titanium are determined differently. The last orbit of a titanium atom has two electrons but its valence electrons are not two. This…

The 21st element in the periodic table is scandium. The element of group-3 is scandium and its symbol is ‘Sc’. Scandium participates in the formation of bonds through its valence electrons. This…

The 20th element in the periodic table is calcium. Calcium is an alkaline earth metal and its symbol is ‘Ca’. Calcium participates in the formation of bonds through its valence electrons. This…
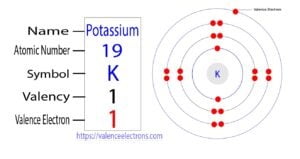
Potassium is the 19th element in the periodic table. Potassium is an alkali metal and its symbol is ‘K’. Potassium forms bonds through its valence electrons. This article discusses in detail how…
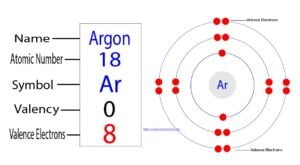
Argon is an inert element and its symbol is ‘Ar’. Argon atoms do not participate in the formation of any bonds. This article discusses in detail how to easily calculate the number…
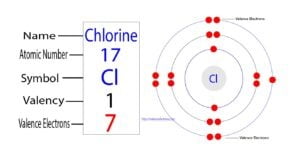
The 17th element of the periodic table is chlorine and it is also an element of group-17. Its symbol is ‘Cl’. Chlorine forms bonds through its valence electrons. What are the valence…
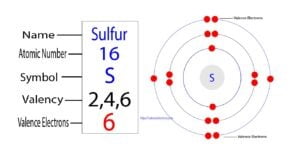
The sixteenth element of the periodic table is sulfur. The element of group-16 is sulfur and its symbol is ‘S’. Sulfur forms bonds through its valence electrons. What are the valence electrons…
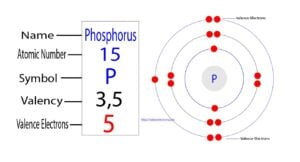
The fifteenth element of the periodic table is phosphorus. The element of group-15 is phosphorus and its symbol is ‘P’. Phosphorus forms bonds through its valence electrons. What are the valence electrons…
The 14th element of the periodic table is silicon. Silicon is a semiconductor material and, its symbol is ‘Si’. Silicon participates in the formation of bonds through its valence electrons. What are…
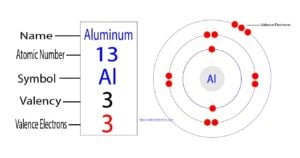
The 13th element of the periodic table is aluminum. The element of group-13 is aluminum and its symbol is Al. Aluminum forms bonds through its valence electrons. What are the valence electrons…
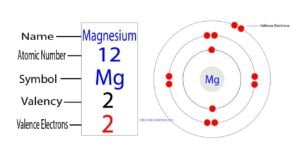
The 12th element in the periodic table is magnesium. Magnesium is an alkaline earth metal and its symbol is ‘Mg’. Magnesium participates in the formation of bonds through valence electrons. What are…
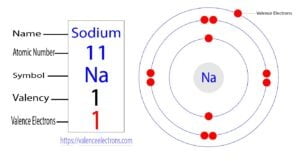
Sodium is the eleventh element in the periodic table. Sodium is an alkali metal and its symbol is ‘Na’. Sodium atoms participate in the formation of bonds through valence electrons. What are…
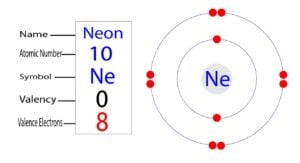
Neon is the tenth element in the periodic table. Neon is an inert element and its symbol is ‘Ne’. Neon atoms do not participate in the formation of any bonds. What are…